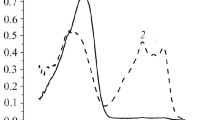
Abstract
1-Ethoxy-2-(2-hydroxy-3-sulfo-5-chlorophenylazo)butane-1,3-dion (L1), 1-ethoxy-2-(2-hydroxy-3,5-disulfophenylazo)butane-1,3-dion (L2), 1-ethoxy-2-(2-hydroxy-4-nitrophenylazo)butane-1,3-dion (L3), and 1-ethoxy-2-(2-hydroxyphenylazo)butane-1,3-dion (L4) were synthesized on the basis of ethyl acetoacetate, and their analytical characteristics were studied. The following dissociation constants of the reagents were determined by potentiometric titration: pK 1 = 5.94 ± 0.04 and pK 2 = 9.75 ± 0.04 (L1), pK 1 = 5.78 ± 0.02 and pK 2 = 9.55 ± 0.03 (L2), pK 1 = 6.03 ± 0.03 and pK 2 = 9.86 ± 0.05 (L3), and pK 1 = 6.19 ± 0.05 and pK 2 = 9.98 ± 0.06 (L4). The chemical analytical characteristics of copper(II) complexes of L1–L4 were determined by spectrophotometry. It was found that, with an increase in the elecrophilic properties of the substituents introduced into the aromatic ring of the molecule, the sensitivity of the reaction increased; the substituents had no effect on the ratio of Cu : L = 1 : 2. A photometric procedure was developed for determining copper in nickel-based alloys.
Similar content being viewed by others
References
Basargin, N.N., Extended Abstract of Doctoral (Chem.) Dissertation, Moscow, 1975.
Korrelyatsii i prognozirovanie analiticheskikh svoistv organicheskikh reagentov i khelatnykh sorbentov (Correlations and Prediction of the Analytical Properties of Organic Reagents and Chelating Polymer Sorbents), Basargin, N.N. and Isaev, E.I., Eds., Moscow: Nauka, 1986.
Gur’eva, R.F. and Savvin, S.B., Zh. Anal. Khim., 2002, vol. 57, no. 11, p. 1158 [J. Anal. Chem. (Engl. Transl.), vol. 57, no. 11, p. 980].
Ivanov, V.M. and Kochelaeva, G.A., Usp. Khim., 2006, vol. 75, no. 3, p. 283.
Savvin, S.B., Shtykov, S.N., and Mikhailova, A.V., Usp. Khim., 2006, vol. 75, no. 4, p. 380.
Ivanov, V.M. and Mamedov, A.M., Zh. Anal. Khim., 2006, vol. 61, no. 11, p. 1128 [J. Anal. Chem. (Engl. Transl.), vol. 61, no. 11, p. 1040].
Makhmudov, K.T., Cand. Sci. (Chem.) Dissertation, Baku, 2006.
Korostelev, P.P., Prigotovlenie rastvorov dlya khimikoanaliticheskikh rabot (Preparation of Solutions for Chemical Analytical Works), Moscow: Khimiya, 1964.
Bates, R.G., Determination of pH: Theory and Practice, New York: Wiley, 1973.
Dyatlova, N.M., Temkina, V.Ya., and Kolpakova, I.D., Kompleksony (Complexones), Moscow: Khimiya, 1970.
Gadzhieva, S.R., Mursalov, T.M., Makhmudov, K.T., and Chyragov, F.M., Koord. Khim., 2006, vol. 32, no. 4, p. 316 [Russ. J. Coord. Chem. (Engl. Transl.), vol. 32, no. 4, p. 304].
Gadzhieva, S.R., Mursalov, T.M., Makhmudov, K.T., Pashaev, F.G., and Chyragov, F.M., Zh. Anal. Khim., 2006, vol. 61, no. 6, p. 598 [J. Anal. Chem. (Engl. Transl.), vol. 61, no. 6, p. 550].
Bulatov, M. and Kalinkin, I., Prakticheskoe rukovodstvo po fotokolorimetricheskim i spektrofotometricheskim metodam analiza (Manual on Photocolorimetric and Spectrophotometric Analytical Techniques), Leningrad: Khimiya, 1972.
Dorokhova, E.N. and Prokhorova, G.V., Zadachi i voprosy po analiticheskoi khimii (Problems and Questions on Analytical Chemistry), Moscow: Mir, 2001.
Astakhov, K.V., Verinikin, V.B., Zimin, V.I., and Zvereva, A.D., Zh. Neorg. Khim., 1961, vol. 6, no. 6, p. 2069.
Podchainova, V.N. and Simonova, L.N., Analiticheskaya khimiya medi (Analytical Chemistry of Copper), Moscow: Nauka, 1990.
Novopol’tseva, V.M. and Osipov, A.K., Zh. Anal. Khim., 2006, vol. 61, no. 10, p. 1067 [J. Anal. Chem. (Engl. Transl.), vol. 61, no. 10, p. 985].
Batuner, L.M. and Pozin, M.E., Matematicheskie metody v khimicheskoi tekhnike (Mathematical Methods in Chemical Engineering), Leningrad: Khim. Lit., 1963.
Author information
Authors and Affiliations
Additional information
Original Russian Text © K.T. Makhmudov, R.A. Alieva, S.R. Gadzhieva, F.M. Chyragov, 2008, published in Zhurnal Analiticheskoi Khimii, 2008, Vol. 63, No. 5, pp. 479–482.
Rights and permissions
About this article
Cite this article
Makhmudov, K.T., Alieva, R.A., Gadzhieva, S.R. et al. Photometric determination of copper(II) in nickel alloys using azoderivatives of ethyl acetoacetate. J Anal Chem 63, 435–438 (2008). https://doi.org/10.1134/S1061934808050055
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934808050055