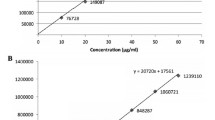
Abstract
Two procedures were proposed for the quantitative analysis of the drug Pentalgin N with the use of HPLC in gradient and isocratic modes. Analgin (dipyrone), caffeine, naproxen, phenobarbital, codeine, an analgin degradation product, and sodium sulfite (added to the test solution to stabilize analgin) were separated on a column (150 × 3.9 mm) packed with Nova-Pak C18 (4.0 μm) with elution with a 0.00625 M KH2 PO4 solution with an acetonitrile concentration gradient from 10 to 60 vol % in 10 min or on a column (150 × 3.9 mm) packed with Nova-Pak CN HP (4.0 μm) with elution with a 0.0110 M KH2 PO4 solution (pH 5.8) containing 5 vol % acetonitrile. The wavelength of the diode-array detector was 212 nm. Model solutions containing all of the active principles and additives of the tablets were analyzed, and the performance characteristics of both procedures were calculated. Both procedures afford reliable analytical results; however, the isocratic version is technically simpler and more preferable for product control in commercial production.
Similar content being viewed by others
References
Vergeichik, E.N. and Onegova, N.S., Farmatsiya, 2001, vol. 50, p. 24.
Larionova, S.G., Dement’eva, N.N., Nechaeva, E.B., Nazarenko, P.V., and Nesterova, G.A., Farmatsiya, 2002, vol. 51, p. 16.
Schoenmakers, P.J., Optimization of Chromatographic Selectivity, Amsterdam: Elsevier, 1986. Translated under the title Optimizatsiya selektivnosti v khromatografii, Moscow: Mir, 1989.
The United States Pharmacopeia, 25th ed., Rockville: U. S. Pharmacopeial Convention, 2001, p. 1991.
Author information
Authors and Affiliations
Additional information
Original Russian Text © G.B. Golubitskii, E.V. Budko, V.M. Ivanov, 2006, published in Zhurnal Analiticheskoi Khimii, 2006, Vol. 61, No. 1, pp. 74–79.
Rights and permissions
About this article
Cite this article
Golubitskii, G.B., Budko, E.V. & Ivanov, V.M. Quantitative analysis of Pentalgin N tablets by gradient and isocratic high-performance liquid chromatography. J Anal Chem 61, 67–71 (2006). https://doi.org/10.1134/S106193480601014X
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1134/S106193480601014X