Abstract
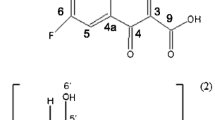
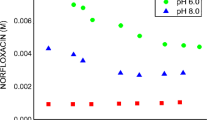
The physicochemical properties and structure of moxifloxacin‒methyl-β-cyclodextrin complex have been studied by UV spectroscopy, FTIR spectroscopy, and computer simulation. The optimal conditions for the formation of the complex have been determined, and the dissociation constant of the complex in acidic media (K dis = (5.0 ± 0.3) × 10–5 М) has been obtained. It has been found that complexation significantly slows down the release of the drug in acidic media. Experimental results are in good agreement with computer simulation data. The following mechanism of complex formation has been proposed: the incorporation of the aromatic fragment of moxifloxacin into the cavity of methyl-β-cyclodextrin is followed by additional stabilization of the complex via multiple hydrophobic interactions and hydrogen bonding.
Similar content being viewed by others
References
O’Donnell, J.A. and Gelone, S.P., Infect. Dis. Clin. North Am., 2000, vol. 14, p. 489.
Chen, T.C., Tsai, T.Y., and Chang, S.W., Exp. Eye Res., 2016, vol. 145, p. 10.
Long, T.E., Keding, L.C., Lewis, D.D., Anstead, M.I., Withers, T.R., and Yu, H.D., Bioorg. Med. Chem. Lett., 2016, vol. 26, p. 1305.
Principi, N. and Esposito, S., Int. J. Antimicrob. Agents, 2015, vol. 45, p. 341.
Jabeen, K., Shakoor, S., and Hasan, R., Int. J. Infect. Dis., 2015, vol. 32, p. 118.
Thee, S., Garcia-Prats, A.J., Donald, P.R., Hesseling, A.C., and Schaaf, H.S., Tuberculosis, 2015, vol. 95, p. 229.
Balabanova, Y., Ignatyeva, O., Fiebig, L., Riekstina, V., Danilovits, M., Jaama, K., Davidaviciene, E., Radiulyte, B., Popa, C.M., Nikolayevskyy, V., and Drobniewski, F., Thorax, 2016, vol. 71, p. 854.
Falzon, D., Jaramillo, E., Schünemann, H.J., et al., Eur. Respir. J., 2011, vol. 38, p. 516.
Van Doorslaer, X., Dewulf, J., Van Langenhove, H., and Demeestere, K., Sci. Total Environ., 2014, vols. 500–501, p. 250.
Padeiskaya, E.N. and Yakovlev, V.P., Antimikrobnye preparaty gruppy ftorkhinolonov v klinicheskoi praktike (Antimicrobial Preparations of Fluoroquinolone Group in Clinical Practice), Moscow: LOGATA, 1998.
Chadha, R., Kashid, N., and Saini, A., J. Sci. Ind. Res., 2014, vol. 63, p. 211.
Li, J. and Zhang, X., J. Incl. Phenom. Macrocycl. Chem., 2011, vol. 69, p. 173.
Del Valle, E.M., Process Biochem., 2004, vol. 34, p. 1033.
http://images.alfresco.advanstar.com/alfresco_images/ pharma/2014/08/22/cc14038c-5022-4d20-b3d2-0bc- 14c095065/article-36186.pdf.
Rodriguez-Aller, M., Guinchard, S., Guillarme, D., Pupier, M., Jeannerat, D., Rivara-Minten, E., Veuthey, J., and Gurny, R., Eur. J. Pharm. Biopharm., 2015, vol. 95, p. 203.
Gharib, R., Greige-Gerges, H., Fourmentin, S., Charcosset, C., and Auezova, L., Carbohydr. Res., 2015, vol. 129, p. 175.
Boogard, M.V., PhD Thesis (University of Groningen), 2003.
Singh, M., Sharma, R., and Banerjee, U.C., Biotechnol. Adv., 2002, vol. 20, p. 341.
Loftsson, T. and Petersen, D.S., Drug Dev. Ind. Pharm., 1998, vol. 24, p. 365.
Szente, L. and Szejtli, J., Adv. Drug Deliv. Rev., 1999, vol. 36, p. 17.
Misiuk, W. and Jozefowicz, M., J. Mol. Liq., 2015, vol. 202, p. 101.
Bani-Yaseen, A.D., Spectrochim. Acta, 2015, vol. 148, p. 93.
Zoete, V., Cuendet, M.A., Grosdidier, A., and Michielin, O., J. Comput. Chem., 2011, vol. 32, p. 2359.
MacKerell, A.D., Jr., Banavali, N., and Foloppe, N., Biopolymers, 2000, vol. 56, p. 257.
Humphrey, W., Dalke, A., and Schulten, K., J. Mol. Graph., 1996, vol. 14, p. 33.
Phillips, J.C., Braun, R., Wang, W., Gumbart, J., Tajkhorshid, E., Villa, E., Chipot, C., Skeel, R.D., Kale, L., and Schulten, K., J. Comput. Chem., 2005, vol. 26, p. 1781.
Koester, L.S., Guterres, S.S., Le Roch, M., Eifler-Lima, V.L., Zuanazzi, J.A., and Bassani, V.L., Drug Dev. Ind. Pharm., 2001, vol. 27, p. 533.
Chao, J., Meng, D., Li, J., Xu, H., and Huang, S., Spectrochim. Acta, 2004, vol. 60, p. 729.
Szekely-Szentmiklosi, B. and Tokes, B., Acta Med. Marisiensis, 2011, vol. 57, p. 116.
Liu, Y. and Yuan, X., J. Incl. Phenom. Macrocycl. Chem., 2015, vol. 82, p. 311.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Skuredina, I.M. Le-Deygen, I.V. Uporov, E.V. Kudryashova, 2017, published in Kolloidnyi Zhurnal, 2017, Vol. 79, No. 5, pp. 627–635.
Rights and permissions
About this article
Cite this article
Skuredina, A.A., Le-Deygen, I.M., Uporov, I.V. et al. A study of the physicochemical properties and structure of moxifloxacin complex with methyl-β-cyclodextrin. Colloid J 79, 668–676 (2017). https://doi.org/10.1134/S1061933X17050143
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X17050143