Abstract
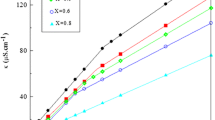
Micellar behavior of binary combinations of ionic liquid, 1-tetradecyl-3-methylimidazolium bromide, with conventional cationic surfactant 1-hexadecylpyridinium bromide was investigated by means of conductometry to study the effect of cosolvent and water content and temperature. The critical micelle concentration and the degree of counterion association were calculated from the conductometry data. Thermodynamic parameters were obtained from the temperature dependence of the critical micelle concentration. The standard Gibbs energy of micellization increased with the increasing percentage of cosolvent as well as the mole fraction of C14mimBr. The standard enthalpy and standard entropy of micelle formation were both decreased with the increasing temperature and the concentration of cosolvent. The entropy contribution was larger than the enthalpic one in pure water, whereas in the ethylene glycol/H2O mixture the enthalpy contribution was predominant
Similar content being viewed by others
References
Li, W.J., Zhang, Z.F., Zhang, J.L., et al., Fluid Phase Equilib., 2006, vol. 248, p. 211.
Martins, M.A.P., Frizzo, C.P., Moreira, et al, Chem. Rev., 2008, vol. 108, p. 2015.
Hapiot, P. and Lagrost, C., Chem. Rev., 2008, vol. 108, p. 2238.
MacFarlane, D.R., Forsyth, M., Howlett, P.C., et al., Acc. Chem. Res., 2007, vol. 40, p. 1165.
Han, X., and Armstrong, D.W., Acc. Chem. Res., 2007, vol. 40, p. 1079.
Zhou, Y. and Antonietti, M., Adv. Mater., 2003, vol. 15, p. 1452.
Welton T. In: Multiphase Homogeneous Catalysis, Ed. by B. Cornils, W.A. Herrmann, I.T. Horvath et al., Chichester: Wiley, 2005, vol. 2, p. 431.
Zhao, H., Xia, S., and Ma, P., J. Chem. Technol. Bio-technol., 2005, vol. 80, p. 1089.
Dietz, M.L. and Stepinski, C., Green Chem., 2005, vol. 7, p. 747.
Byrne, N., Howlett, C., MacFarlane, R., and Forsyth, M., Adv. Mater., 2005, vol. 17, p. 2497.
Wang, P., Zakeeruddin, M., Moser, J.-E., and Graetzel, M., J. Phys. Chem. B, 2003, vol. 107, p. 13280.
Yamanaka, N., Kawano, R., Kubo, W., et al., Chem. Commun., 2005, p. 740.
Irimescu, R. and Kato, K., J. Mol. Catal. B., 2004. vol. 30, p. 189.
Luczak, J., Hupka, J., Thoming, J., and Jungnickel, C., Colloids Surf. A., 2008, vol. 329. 125.
Miskolczy, Z., Sebok-Nagy, K., Biczok, L., and Gokturk, S., Chem. Phys. Lett., 2004, vol. 400, p. 296.
Beesley, A.H., Evans, D.F., and Laughlin, R.G., J. Phys. Chem. B, 1998 vol. 92, p. 791.
Feng, Q.Q., Wang, H.Y., Zhang, S.B., and Wang, J.J., Colloids Surf. A, 2010, vol. 367, p. 7.
Yan, J., Wang, D., Fang, B., and Yang, F.F., J. Solution Chem., 2010, vol. 39, p. 1501.
Bakshi, M.S., J. Chem. Soc., Faraday Trans., 1997, vol. 93, p. 4005.
Shinoda, K. and Nomura, T., J. Phys. Chem., 1980, vol. 84, p. 365.
Funasaki, N. and Hada, S., J. Colloid Interface Sci., 1980, vol. 84, p. 736.
Illner, G.C. and Hofmann, H., Tenside Surfact. Det., 1995, vol. 32, p. 318.
Tiwari, A.K., Sonu, Saha, S.K., J. Chem. Thermodyn., 2013, vol. 60, p. 29.
Akba, H. and Kartal, C., Colloid J., 2006, vol. 68, p. 125.
Akba, H. and Batşoc, C., Colloid J., 2008, vol. 70, p. 127.
Mukai, T., Yoshio, M., Kato, T., Yoshizawa, M., and Ohno, H., Chem Commun., 2005, vol. 10, p. 1333.
Wasserscheid, P. and Welton, T., Ionic Liquids in Synthesis, Weinheim: Wiley-VCH, 2002.
Chakraborty, T., Ghosh, S., and Moulik, S.P., J. Phys. Chem. B., 2005, vol. 109, p. 14813.
Ghosh, S. and Chakraborty, T., J. Phys. Chem. B, 2007, vol. 111, p. 8080.
Chakraborty, T. and Ghosh, S., Colloid Polym. Sci., 2007, vol. 285, p. 1665.
Ghosh, S., J. Colloid Interface Sci., 2001, vol. 244, p. 128.
Bakshi, M.S. and Kaur, G., J. Mol. Liq., 2000, vol. 88, p. 15.
Lianos, P. and Lang, J., J. Colloid Interface Sci., 1983 vol. 96, p. 222.
Das, C., Chakraborty, T., Ghosh, S., and Das, B., Colloid Polym. Sci., 2008, vol. 286, p. 1143.
Carnero Ruiz, C., Molina-Bolivar, J.A., Aguiar, J., et al., Colloid Polym. Sci., 2003, vol. 281, p. 531.
Ling, J.L., Xu, M.H., Hong, D., and Zhang, Y., Chem. Res. A, 2012, vol. 2, p. 322.
Wasserscheid, P. and Welton, T., Ionic Liquids in Synthesis, Weinheim: Wiley–VCH, 2002.
Chakraborty, T., Ghosh, S., and Moulik, S.P., J. Phys. Chem. B., 2005, vol. 109, p. 14813.
Ghosh, S. and Chakraborty, T., J. Phys. Chem. B, 2007, vol. 111, p. 8080.
Chakraborty, T. and Ghosh, S., Colloid Polym. Sci., 2007, vol. 285, p. 1665.
Ghosh, S., J. Colloid Interface Sci., 2001, vol. 244, p. 128.
Bakshi, M.S. and Kaur, G., J. Mol. Liq., 2000, vol. 88, p. 15.
Lianos, P. and Lang, J., J. Colloid Interface Sci., 1983 vol. 96, p. 222.
Das, C., Chakraborty, T., Ghosh, S., and Das, B., Colloid Polym. Sci., 2008, vol. 286, p. 1143.
Carnero Ruiz, C., Molina-Bolivar, J.A., Aguiar, J., et al., Colloid Polym. Sci., 2003, vol. 281, p. 531.
Ling, J.L., Xu, M.H., Hong, D., and Zhang, Y., Chem. Res. A, 2012, vol. 2, p. 322.
Callaghan, A., Doyle, R., and Alexander, E., Langmuir, 1993, vol. 9, p. 3422.
Ruiz, C.C., Colloid Polym. Sci., 1999, vol. 277, p. 701.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
He, Y., Sun, L., Fang, D. et al. Aggregation behaviour and thermodynamics of mixed micellization of 1-hexadecylpyridinium bromide and ionic liquid in ethylene glycol/water binary mixtures. Colloid J 76, 96–103 (2014). https://doi.org/10.1134/S1061933X14010062
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X14010062