Abstract
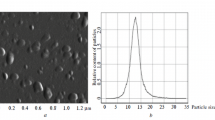
The structure and adsorption properties (with respect to water vapor and nickel cations) of chitosan samples dehydrated by either lyophilization after precipitation with NaOH or Na2CO3 or drying in air are investigated. IR spectroscopy and X-ray diffraction data testify that the lyophilization causes both changes in the conformation of chitosan macromolecules and a substantial rise in the crystallinity of the polymer to compare with the air dried one. Chitosan structure ordering resultant from the lyophilization noticeably increases its sorption ability with respect to both water vapor and nickel cations. For example, the sorption capacity of the lyophilized chitosan with respect to nickel cations is as high as 4.5 mmol/g and, thus, is threefold higher than that of air-dried chitosan. The comparative analysis of water adsorption isotherms in the region of low degrees of filling indicates that chitosan structure ordering resultant from the lyophilization changes the number and the energy of primary adsorption sites.
Similar content being viewed by others
References
Smirnov, A.D., Sorbtsionnaya ochistka vody (Sorption Water Treatment), Leningrad: Khimiya, 1982.
Juang, R.-S., Wu, F.-C., and Tseng, R.-L., Water Res., 1999, vol. 33, p. 2403.
Krasil’nikova, O.K., Artamonova, S.D., Vikhoreva, G.A., et al., Abstracts of Papers, VI Mezhdunar. konf. “Novye dostizheniya v isledovanii khitina i khitozana” (VI Int. Conf. “Progress in Investigation of Chitin and Chitosan”), Shchelkovo, 2001, p. 295.
Schwarzenbach, G. and Flaschka, H., Die komplexometrische Titration, Stuttgart: Ferdinand Enke, 1965.
Vartapetyan, R.Sh. and Voloshchuk, A.M., Usp. Khim., 1995, vol. 64, p. 1055.
Ratto, J., Hatakeyama, T., and Blumsteun, R.B., Polymer, 1995, vol. 36, p. 2915.
Krasilnikova, O.K. and Vartapetian, R.Sh., Abstracts of Papers, 5 Int. Conf. on Polymer-Solvent Complexes & Intercalates, Lorient, France, 2004, p. 13.
Krasil’nikova, O.K., Sarylova, M.E., Volkov, A.V., et al., Vysokomol. Soedin., Ser. A, 1991, vol. 3, p. 202.
Juang, R.-S. and Shao, H.-J., Water Res., 2002, vol. 36, p. 2999.
Dubinin, M.M., Izv. Akad. Nauk SSSR, Ser. Khim., 1974, no. 5, p. 996.
Stoeckli, F., Lopez-Ramon, V.M., and Moreno-Castilla, C., Langmuir, 2001, vol. 17, p. 3301.
Stadnik, A.M. and El’tekov, Yu.A., Zh. Prikl. Khim., 1975, vol. 49, p. 666.
Kharitonova, A.G., Krasil’nikova, O.K., Vartapetyan, R.Sh., and Bulanova, A.V., Kolloidn. Zh., 2005, vol. 67, p. 416.
Ding, Ch., Song, Q., and Ye, S., J. East China Univ. Sci. Technol., 2003, vol. 29, p. 6.
Yuan, Y.-C., Zhang, M.-Q., and Rong, M.-Z., Huaxue Xuebao, Acta Chimica Cinica, 2005, vol. 63, p. 1753.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.V. Solovtsova, T.Yu. Grankina, O.K. Krasil’nikova, N.V. Serebryakova, S.M. Shinkarev, A.M. Voloshchuk, 2008, published in Kolloidnyi Zhurnal, 2008, Vol. 70, No. 3, pp. 376–383.
Rights and permissions
About this article
Cite this article
Solovtsova, O.V., Grankina, T.Y., Krasil’nikova, O.K. et al. The effect of the dehydration conditions of chitosan-based polymeric adsorbents on the adsorption of nickel cations. Colloid J 70, 341–348 (2008). https://doi.org/10.1134/S1061933X08030125
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X08030125