Abstract
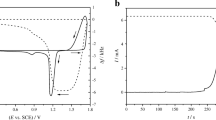
The influence of manganese ions on the electrodeposition process of lead dioxide on the glassy carbon electrode (GCE) in the lead nitrate solution was investigated by cyclic voltammetry (CV), chronoamperometry (CA) and electrochemical impedance spectroscopy (EIS). The results show that the electrodeposition process of lead dioxide was inhibited by the addition of manganese ions. However, the nucleation model is not influenced by the addition of manganese ions and the electrodeposition process still fits the instantaneous nucleation model with three-dimensional growth according to Scharifker—Hills’ model.
Similar content being viewed by others
References
Ramirez, G., Recio, F.J., Herrasti, P., Ponce-de-Leon, C., and Sires, I., Effect of RVC porosity on the performance of PbO2 composite coatings with titanate nano-tubes for the electrochemical oxidation of azo dyes, Electrochim. Acta, 2016, vol. 204, p. 9.
Amadelli, R., Samiolo, L., Battisti, A., and Velichenko, A.B., Electro-oxidation of some phenolic compounds by electrogenerated O3 and by direct electrolysis at PbO2 anodes, J. Electrochem. Soc., 2011, vol. 158, p. P87.
Egan, D.R.P., Low, C.T.J., and Walsh, F.C., Electro-deposited nanostructured lead dioxide as a thin film electrode for a lightweight lead-acid battery, J. Power Sources, 2011, vol. 196, p. 5725.
Shmychkova, O., Luk’yanenko, T., Yakubenko, A., Amadelli, R., and Velichenko, A., Electrooxidation of some phenolic compounds at Bi-doped PbO2, Appl. Catal. B-Environ, 2015, vol. 162, p. 346.
Dai, Q.Z., Zhou, J.Z., Weng, M.L., Luo, X.B., Feng, D.L., and Chen, J.M., Electrochemical oxidation metronidazole with Co modified PbO2 electrode: degradation and mechanism, Sep. Purif. Technol., 2016, vol. 166, p. 109.
Andrade, L.S., Ruotolo, L.A.M., Rocha-Filho, R.C., Bocchi, N., Biaggio, S.R., Iniesta, J., Garcia-Garcia, V., and Montiel, V., On the performance of Fe and Fe,F doped Ti–Pt/PbO2 electrodes in the electrooxidation of the Blue Reactive 19 dye in simulated textile waste-water, Chemosphere, 2007, vol. 66, p. 2035.
Xu, M., Wang, Z.C., Wang, F.W., Hong, P., Wang, C.Y., Ouyang, X.M., Zhu, C.G., Wei, Y.J., Hun, Y.H., and Fang, W.Y., Fabrication of cerium doped Ti/nano-TiO2/PbO2 electrode with improved electrocatalytic activity and its application in organic degradation, Electrochim. Acta, 2016, vol. 201, p. 240.
Xu, H., Shao, D., Zhang, Q., Yang, H.H., and Yan, W., Preparation and characterization of PbO2 electrodes from electro-deposition solutions with different copper concentration, RSC Adv., 2014, vol. 4, p. 25011.
Dalili, N., Clark, M.P., Davari, E., and Ivey, D.G., Microstructural characterization of the cycling behavior of electrodeposited manganese oxide supercapacitors using 3D electron tomography, J. Power Sources, 2016, vol. 328, p. 318.
Li, Y.J., Wang, G.L., Ye, K., Cheng, K., Pan, Y., Yan, P., Yin, J.L., and Cao, D.X., Facile preparation of three-dimensional multilayer porous MnO2/reduced graphene oxide composite and its supercapacitive performance, J. Power Sources, 2014, vol. 271, p. 582.
Li, X.H. and Pletcher, D., Electrodeposited lead dioxide coatings, Chem. Soc. Rev., 2011, vol. 40, p. 3879.
Li, Y., Jiang, L.X., Liu, F.Y., Li, J., and Liu, Y.X., Novel phosphorus-doped PbO2–MnO2 bicontinuous electrodes for oxygen evolution reaction, RSC Adv., 2014, vol. 4, p. 24020.
Comisso, N., Cattarin, S., Guerriero, P., Mattarozzi, L., Musiani, M., and Verlato, E., Conversion of porous Mn2+ ions, Electrochem. Commun., 2016, vol. 73, p. 59.
Yang, H.T., Chen, B.M., Liu, H.R., Guo, Z.C., Zhang, Y.C., Li, X.L., and Xu, R.D., Effects of manganese nitrate concentration on the performance of an aluminum substrate ß-PbO2–MnO2–WC–ZrO2 composite electrode material, Int. J. Hydrogen Energy, 2014, vol. 39, p. 3087.
Li, P., Zhao, Y.M., Ding, B.B., and Wang, L.Z., Effect of calcination temperature and molar ratio of tin and manganese on capacitance of Ti/SnO2–Sb–Mn/ß–PbO2 electrode during phenol electro-oxidation, J. Electroanal. Chem., 2015, vol. 747, p. 45.
Dan, Y.Y., Lin, H.B., Liu, X.L., Lu, H.Y., Zhao, J.Z., Shi, Z., and Guo, Y.P., Porous quasi three-dimensional nano-Mn3O4 + PbO2 composite as supercapacitor electrode material, Electrochim. Acta, 2012, vol. 83, p. 175.
Abyaneh, M.Y., Saez, V., Gonzalez-Garcia, J., and Mason, T.J., Electrocrystallization of lead dioxide: analysis of the early stages of nucleation and growth, Electrochim. Acta, 2010, vol. 55, p. 3572.
Hwang, B.J., Santhanam, R., and Chang, Y.W., Mechanism of electrodeposition of PbO2 at a Pt sheet/rotating disk electrode, Electroanalysis, 2002, vol. 14, p. 5363.
Velichenko, A.B., Girenko, D.V., and Danilov, F.I., Electrodeposition of lead dioxide at an Au electrode, Electrochim. Acta, 1995, vol. 40, p. 2803.
Gonzalez-Garcia, J., Iniesta, J., Exposito, E., GarciaGarcia, V., Montiel, V., and Aldaz, A., Early stages of lead dioxide electrodeposition on rough titanium, Thin Solid Films, 1999, vol. 352, p. 49.
Gonzalez-Garcia, J., Gallud, F., Iniesta, J., Montiel, V., Aldaz, A., and Lasia, A., Kinetics of electrocrystallisation of PbO2 on glassy carbon electrodes: influence of ultrasound, New J. Chem., 2001, vol. 25, p. 1195.
Saez, V., Gonzalez-Garcia, J., Iniesta, J., Frias-Ferrer, A., and Aldaz, A., Electrodeposition of PbO2 on glassy carbon electrodes: influence of ultrasound frequency, Electrochem. Commun., 2004, vol. 6, p. 757.
Velichenko, A.B., Amadelli, R., Knysh, V.A., Luk’yanenko, T.V., and Danilov, F.I., Kinetics of lead dioxide electrodeposition from nitrate solutions containing colloidal TiO2, J. Electroanal. Chem., 2009, vol. 632, p. 192.
Lin, Z.B., Xie, B.G., Chen, J.S., Sun, J.J., and Chen, G.N., Nucleation mechanism of silver nucleation mechanism of silver during electrodeposition on a glassy carbon electrode from a cyanide-free bath with 2-hydroxypyridine as a complexing agent, J. Electroanal. Chem., 2009, vol. 633, p. 207.
Yao, Y.W., Zhou, T., Zhao, C.M., Jing, Q.M., and Wang, Y., Influence of ZrO2 particles on fluorine-doped lead dioxide electrodeposition process from nitrate bath, Electrochim. Acta, 2013, vol. 99, p. 225.
Scharifker, B. and Hills, G., Theoretical and experimental studies of multiple nucleation, Electrochim. Acta, 1983, vol. 28, p. 879.
Gu, M., Initial stages of the electrocrystallization of Co-Cu alloys on GCE from the Co rich electrolytes, Electrochim. Acta, 2007, vol. 52, p. 4443.
Hu, F. and Chan, K.C., Equivalent circuit modelling of Ni-SiC electrodeposition under ramp-up and ramp-down waveforms, Mater. Chem. Phys., 2006, vol. 99, p. 424.
Pasquale, M.A., Gassa, L.M., and Arvia, A.J., Copper electrodeposition from an acidic plating bath containing accelerating and inhibiting organic additives, Electrochim. Acta, 2008, vol. 53, p. 5891.
Aaboubi, O., Douglade, J., Abenaqui, X., Boumedmed, R., and Vonhoff, J., Influence of tartaric acid on zinc electrodeposition from sulphate bath, Electrochim. Acta, 2011, vol. 56, p. 7885.
Shmychkova, O., Lukyanenko, T., Velichenko, A., Meda, L., and Amadelli, R., Bi-doped PbO2 anodes: electrodeposition and physico-chemical properties, Electrochim. Acta, 2013, vol. 111, p. 332.
Shmychkova, O., Lukyanenko, T., Amadelli, R., and Velichenko, A., Electrodeposition of Ce-doped PbO2. J. Electroanal. Chem., 2013, vol. 706, p. 86.
Velichenko, A.B., Baranova, E.A., Girenko, D.V., Amadelli, R., Kovalev, S.V., and Danilov, F.I., Mechanism of electrodeposition of lead dioxide from nitrate solutions, Russ. J. Electrochem., 2003, vol. 39, p. 615.
Velichenko, A.B. and Devilliers, D., Electrodeposition of fluorine-doped lead dioxide, J. Fluorine Chem., 2007, vol. 128, p. 269.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Russian in Elektrokhimiya, 2019, Vol. 55, No. 5, pp. 529–534.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Yao, Y., Huang, C., Dong, H. et al. Influence of Manganese Ions on the Electrodeposition Process of Lead Dioxide in Lead Nitrate Solution. Russ J Electrochem 55, 364–369 (2019). https://doi.org/10.1134/S1023193519040049
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193519040049