Abstract
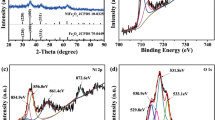
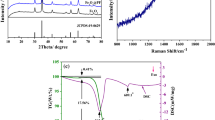
The Nanometer and micrometer sized samples of FeNbO4 were prepared by sol-gel polymeric precursor and solid state reaction methods. Electrochemical lithium insertion was performed into both samples. Structural changes during the lithium insertion were followed by ex-situ XRD. The electrochemical charge discharge curves shows insertion of 0.3 Li is obtained in micrometer sized samples when discharged to 1.0 V and extraction of 0.1 Li is observed during charge. On the other hand, in the nano structured sample, insertion of 3Li/FeNbO4 were observed and during the first discharge, 2.2Li is extracted when charged to 3 V. A reversible capacity of 120 mA h/g is observed after 20 cycles.
Similar content being viewed by others
References
Dawson, D.H. and Williams, D.E., As-sensitive resistors: surface interaction of chlorine with semiconducting oxides, J. Mater. Chem., 1996, vol. 6, p. 409.
Leiva, H., Sieber, K., Khazai, B., Dwight, K., and Wold, A., A structural and electronic relationships between conducting iron niobates and iron tungstates, J. Solid State Chem., 1982, vol. 44, p. 113.
Ehrenberg, H., Wltschek, G., and Theissmann, R., The Magnetic structure of FeNbO4, J. Magn. Mater., 2000, vol. 218, p. 261.
Schmidbauer, E., Electrical conductivity, thermopower 57Fe Mossbauer spectroscopy of solid solutions Fe(Nb1− xWx)O4, 0 ≤ x ≤ 0.4, J. Phys. Condens. Matter, 1998, vol. 10, p. 8279.
Roth, R.S. and Waring, J.L., Ixiolite and other polymorphic types of FeNbO4, Amer. Miner., 1964, vol. 49, p. 140.
Harrison, W.T.A. and Cheetham, A.K., Structural and magnetic properties of FeNbO4-II, Mater. Res. Bull., 1989, vol. 24, p. 523.
Koenitzer, J., Khazai, B., and Hormadaly, J., Preparation and photoelectronic properties of FeNbO4, J. Solid State Chem., 1980, vol. 35, p. 128.
Noda, Y., Shimada, M., Koizumi, M., and Kanamaru, F., Magnetic and electrical properties and Mossbauer effect in the solid solution Fe(Nb1 − xWx)O4 (0 ≤ x ≤ 1), J. Solid State Chem., 1979, vol. 28, p. 379.
Theissmann, R., Ehrenberg, H., Weitzel, H., and Fuess, H., Nanostructured cation distribution in FeNbO4: a synchrotron powder diffraction and transmission electron microscopy investigation, J. Mater. Sci., 2002, vol. 37, p. 4431.
Schmidbauer, E. and Schneider, J., Electrical resistivity, thermo power and 57Fe Mossbauer study of FeNbO4, J. Solid State Chem., 1997, vol. 134, p. 253.
Shim, H.W., Cho, I.S., and Hong, K.S., Hydrothermal synthesis and electrochemical properties of FeNbO4 nano spheres, J. Ceram. Soc. Jpn., 2012, vol. 120, p. 82.
Sri Devi Kumari, T., Vinith Gandhi, R., and Rahul, G., Electrochemical lithium insertion behavior of FeNbO4: structural relations and in situ conversion into FeNb2O6 during carbon coating, Mater. Chem. Phys., 2014, vol. 145, p. 425.
Larcher, D., Masquelier, C., and Bonnin, D., Effect of particle size on lithium intercalation into α-Fe2O3, J. Electrochem. Soc., 2003, vol. 150, p. A133.
Anji Reddy, M., Satya Kishore, M., and Pralong, V., Room temperature synthesis and Li insertion into nanocrystalline rutile TiO2, Electrochem. Commun., 2006, vol. 8, p. 1299.
Zhou, Y., Zifeng Qiu, and Mengkai Lu, Preparation and characterization of porous Nb2O5 nanoparticles, Mater. Res. Bull., 2008, vol. 43, p. 1363.
Anji Reddy, M., Pralong, V., and Varadaraju, U.V., Crystallite size constraints on lithium insertion into brookite TiO2, Electrochem. Solid State Lett., 2008, vol. 11, no. 8, p. A132.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Elektrokhimiya, 2019, Vol. 55, No. 3, pp. 300–307.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Saritha, D. Synthesis, Characterization and Electrochemical Analysis of FeNbO4 Phase with α-PbO2 Structure. Russ J Electrochem 55, 168–174 (2019). https://doi.org/10.1134/S1023193519020071
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193519020071