Abstract
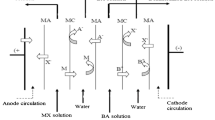
During the electrodialysis of ammonium nitrate solution, the fluxes of salt ions pass through the maximum, which is observed near the limiting current, with increasing current density. A decrease in the flux of ammonium ions at the overlimiting current densities is caused by the effect of competitive transport of solution ions and by the formation of weak NH3 ∙ H2O electrolyte due to the alkalization of solution layer adjacent to the cation-exchange membrane in the desalination channel. A decrease in the flux of nitrate ions in the overlimiting current modes is caused by a change in the composition and catalytic activity of the functional groups of anion-exchange membrane towards the dissociation of water molecules due to the effect of ammonium ions.
Similar content being viewed by others
References
Niftaliev, S.I., Kozaderova, O.A., Kim, K.B., and Malyavina, Yu.M., Electrodialysis in purification of nitrogen-containing sewage of factory for mineral fertilizer production, Khim. Promyshl. Segodnya, 2014, no. 7, p. 52.
Zabolotskii, V.I., Shel’deshov, N.V, and Gnusin N.P., Dissociation of water molecules in systems with ion-exchange membranes, Russ. Chem. Rev., 1988, vol. 57, p. 801.
Shaposhnik, V.A. and Kozaderova, O.A., Transport of hydrogen and hydroxyl ions through ion-exchange membranes under overlimiting current densities, Rus. J. Electrochem., 2012, vol. 48, p. 791.
Pismenskaya, N., Nikonenko, N., Volodina, E., and Pourcelly, G., Electrotransport of weak-acid anions through anion-exchange membranes, Desalination, 2002, vol. 147, p. 345.
Nikonenko, V., Lebedev, K., Manzanares, J.A., and Pourcelly, G., Modelling the transport of carbonic acid anions through anion-exchange membranes, Electrochim. Acta, 2003, vol. 48, p. 3639.
Nefedova, G.Z. and Klimova, Z.G., Ionitovye membrany. Granulyaty. Poroshki: katalog (Ionite Membranes. Granules. Powders: Catalog), Moscow: NIITEKhim, 1977.
Vasiliev, V.P., Kochergina, L.A., and Morozova, R.P., Analiticheskaya Khimiya. Laboratornyi praktikum (Analytical Chemistry. Laboratory Practical Course), Moscow: Drofa, 2006.
NH4 --selektivnyi elektrod serii “Vol’ta”. Pasport i rukovodstvo po ekspluatatsii (NH4 --Selective Electrode of Volta Series. Passport and Manual), St.-Petersburg, 2011.
K +-selektivnyi elektrod serii “Vol’ta”. Pasport i rukovodstvo po ekspluatatsii (K +-Selective Electrode of Volta Series. Passport and Manual), St.-Petersburg, 2011.
NO3 --selektivnyi elektrod serii “Vol’ta”. Pasport i rukovodstvo po ekspluatatsii (NO3 --Selective Electrode of Volta Series. Passport and Manual), St.-Petersburg, 2011.
Rosenberg, N.W. and Tirrel, C.E., Limiting currents in membrane cells, Ind. Eng. Chem., 1957, vol. 49, no. 4, p. 780.
Isaev, N.I. and Shaposhnik, V.A., Dissolution of difficultly soluble electrolytes by electrodialysis with ionite membranes, in Synthesis and Properties of Ion-Exchange Materials, Moscow: Nauka, 1968, p. 256.
Kastyuchik, A.S., Kozaderova, O.A., and Shaposhnik, V.A., Generation of OH– ions at the anion-exchange membrane— solution interface in electrodialysis, Sorpts. Khromatogr. Proc., 2007, vol. 7, no. 5, p. 800.
Shaposhnik, V.A., Kastyuchik, A.S., and Kozaderova, O.A., Irreversible dissociation of water molecules on the ion-exchange membrane—electrolyte solution interface in electrodialysis, Rus. J. Electrochem., 2008, vol. 44, p. 1073.
Shaposhnik, V.A., Vasil’eva, V.I., and Grigorchuk, O.V., Yavleniya perenosa v ionoobmennykh membranakh (Transport Phenomena in Ion-Exchange Membranes), Moscow: MFTI, 2001.
Volkov A.I. and Zharskii, I.M., Bol’shoi khimicheskii spravochkik (Great Handbook of Chemistry), Minsk: Sovremennaya Shkola, 2005.
Shaposhnik, V.A., Selemenev, V.F., and Terent’eva, N.P., The barrier effect during electromigration of proline and valine through ion-exchange membranes in electrodialysis, Zh. Prikl. Khim., 1988, vol. 61, no. 5, p. 1185.
Eliseeva, T.V., Krisilova, E.V., Vasilevskii, V.P., and Novitskii, E.G., Features of electrodialysis of solutions of tartaric acid and its salts, Membr. Membr. Tekhnol., 2012, vol. 2, no. 3, p. 173.
Aminov, O.V., Shaposhnik, V.A., Guba, A.A., and Kutsenko, A.E., Conjugated transport of ammonium ions with hydrogen and hydroxyl ions, Sorpts. Khromatogr. Proc., 2013, vol. 13, no. 6, p. 816.
Tulupov, P.E., Stoikost’ ionoobmennykh materialov (Resistance of Ion-Exchange Materials), Moscow: Khimiya, 1984.
Saldadze, K.M. and Kopylova-Valova, V.D., Kompleksoobrazuyushchie ionity (compleksity) (Complexing Ionites (Complexites)), Moscow: Khimiya, 1980.
Medyantseva, D.G., and Shishkina, S.V., Electrodialysis of nitrate solutions, Izv. Vyssh. Uchebn. Zaved., The North Caucasus region. Natural Sciences, 2008, p. 94.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.A. Kozaderova, S.I. Niftaliev, K.B. Kim, 2018, published in Elektrokhimiya, 2018, Vol. 54, No. 4, pp. 416–422.
Rights and permissions
About this article
Cite this article
Kozaderova, O.A., Niftaliev, S.I. & Kim, K.B. Ionic Transport in Electrodialysis of Ammonium Nitrate. Russ J Electrochem 54, 363–367 (2018). https://doi.org/10.1134/S1023193518040043
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193518040043