Abstract
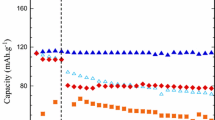
New compositions of liquid organic electrolytes with working temperatures of up to–50°С were developed for low-temperature primary Li/CF x power sources. Five different compositions of organic electrolytes with a 15-crown-5 (2 vol %) addition and without it were studied on laboratory Li/CF x power sources. 1МLiBF4 (LiPF6) in an ethylene carbonate/dimethyl carbonate/methyl propionate/ethylmethyl carbonate (EC/DMC/MP/EMC) (1: 1: 1: 2) mixture and 1 М LiPF6 in an EC/DMC/EMC (1: 1: 3) mixture each with a 15-crown-5 (2 vol %) addition were found to be the best compositions of organic electrolytes with working temperatures of up to–50°С. The electrochemical tests at 20 and–50°С in the Li/CF x system showed that the 15-crown-5 addition increased the length of the discharge plateau at–50°С three- or fourfold. The mechanisms responsible for the increase in the discharge capacity of the CF x cathode in the presence of a crown ether addition were suggested.
Similar content being viewed by others
References
Dey, A.N. and Sallivan, B.P., J. Electrochem. Soc., 1970, vol. 117, p. 222.
Foos, J. S. and Stolki, T. J., J. Electrochem. Soc., 1988, vol. 135, p. 2769.
Geronov, Y., Puresheva, B., Moshtev, R.V., Zlatilova, P., Kosev, T., Staynov, Z., Pistoia, G., and Pasquali, M., J. Electrochem. Soc., 1990, vol. 137, p. 3338.
Dampier, F.W., J. Electrochem. Soc., 1981, vol. 128, p. 2501.
Abraham, K.M., Pasquariello, D.M., and Martin, F.J., J. Electrochem. Soc., 1986, vol. 133, p. 661.
Xu, K., Chem. Rev., 2014, vol. 114, p. 11503.
Linden, D. and Reddy, T.B., Handbook of Batteries, New York: McGraw-Hill, 2002.
Smart, M.C., Ratnakumar, B.V., Surampudi, S., Wang, Y., Zhang, X., Greenbaum, S.G., Hightower, A., Ahn, C.C., and Fultz, B., J. Electrochem. Soc., 1999, vol. 146, p. 3963.
Smart, M.C., Ratnakumar, B.V., and Surampudi, S., J. Electrochem. Soc., 1999, vol. 146, p. 486.
Ohta, A.H., Koshina, H., Okuno, H., and Murai, H., J. Power Sources, 1995, vol. 54, p. 6.
Aurbauch, D. and Granot, E., Electrochim. Acta, 1997, vols. 42–44, p. 697.
Salomon, M. and Hefter, J.T., Pure Appl. Chem., 1993, vol. 65, p. 1533.
D’Aprano, A., Salomon, M., and Mauro, V., J. Solution Chem., 1995, vol. 24, p. 685.
Yarmolenko, O.V. and Tulibaeva, G.Z., Al’tern. Energ. Ekol., 2013, nos. 01–1(117), p. 60.
Yarmolenko, O.V. and Efimov, O. N., Russ. J. Electrochem., 2005, vol. 41, p. 568.
Yarmolenko, O.V., Tulibaeva, G.Z., Khatmullina, K.G., Bogdanova, L.M., and Shestakov, A.F., Mendeleev Commun., 2016, vol. 26, p. 407.
Kawamura, T., Okada, S., and Yamaki, J.-I., J. Power Sources, 2006, vol. 156, p. 547.
Yarmolenko, O.V., Tulibaeva, G.Z., Petrova, G.N., Shuvalova, N.I., and Efimov, O.N., Materialy dokladov 9-ogo mezhdunarodnogo soveshchaniya “Fundamental’nye problemy ioniki tverdogo tela” (Papers from the 9th Int. Conf. Fundamental Problems of Solid State Ionics), Chernogolovka, Granitsa, 2008, p. 204.
Fateev, S.A., Nikol’skaya, N.F., and Polyakova, N.V., Abstracts of Papers, Rossiiskaya konf. “Fizikokhimicheskie problemy vozobnovlyaemoi energetiki” (Russian Conf. “Physicochemical Problems of Renewable Energy”), St. Petersburg: Polytechnical Univ., 2013, p. 118.
Perdew, P., Burke, K., and Ernzerhof, M., Phys. Rev. Lett., 1996, vol. 77, p. 3865.
Laikov, D.N., Chem. Phys. Lett., 1997, vol. 281, p. 151.
Yazami, R., Patent WO2008105916A2, 2007.
Whitacre, J.F., West, W.C., Smart, M.C., Yazami, R., Prakash, G.K.S., Hamwi, A., and Ratnakumar, B.V., Electrochem. Solid-State Lett., 2007, vol. 10, p. A166.
Nagasubramanian, G. and Sanchez, B., J. Power Sources, 2007, vol. 165, p. 630.
Valerga, A.J., Badachhape, R.B., Parks, G.D., Kamarchick, P., Wood, J.L., and Margrave, J.L., U.S. Army Electronics Command, R&D Technical Report, ECOM-0056-F, 1974.
Whittingham, M.S., J. Electrochem. Soc., 1975, vol. 122, p. 526.
Watanabe, N., Solid State Ionics, 1980, vol. 1, p. 87.
Touhara, H., Fujimoto, H., Watanabe, N., and Tressaud, A., Solid State Ionics, 1984, vol. 14, p. 163.
Touhara, H., Fujimoto, H., Kadono, K., Watanabe, N., and Endo, M., Electrochim. Acta, 1987, vol. 32, p. 293.
Zhang, S.S., Foster, D., Wolfenstine, J., and Read, J., J. Power Sources, 2009, vol. 187, p. 233.
Watanabe, N., Hagiwara, R., Nakajima, T., Touhara, H., and Ueno, K., Electrochim. Acta, 1982, vol. 27, p. 1615.
Giraudet, J., Delabarre, C., Guerin, K., Dubois, M., Masin, F., and Hamwi, A., J. Power Sources, 2006, vol. 158, p. 1365.
Abraham, K.M. and Pasquariello, D.M., The Electrochem. Soc. Proceedings, Abraham, K.M. and Salomon, M., Eds., 1991, PV91-3.
Tulibaeva, G.Z., Yarmolenko, O.V., and Shestakov, A.F., Russ. Chem. Bull. (Int. Ed.), 2009, vol. 58, p. 1589.
Yarmolenko, O V., Khatmullina, K.G., Tulibaeva, G.Z., Bogdanova, L.M., and Shestakov, A.F., Russ. Chem. Bull., 2012, vol. 61, p. 539.
Ignatova, A.A., Yarmolenko, O.V., Tulibaeva, G.Z., Shestakov, A.F., and Fateev, S.A., J. Power Sources, 2016, vol. 309, p. 116.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Ignatova, G.Z. Tulibaeva, O.V. Yarmolenko, S.A. Fateev, 2017, published in Elektrokhimiya, 2017, Vol. 53, No. 3, pp. 330–339.
Rights and permissions
About this article
Cite this article
Ignatova, A.A., Tulibaeva, G.Z., Yarmolenko, O.V. et al. Electrolyte systems for primary lithium-fluorocarbon power sources and their working efficiency in a wide temperature range. Russ J Electrochem 53, 292–301 (2017). https://doi.org/10.1134/S1023193517030077
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193517030077