Abstract
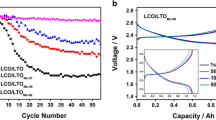
The effect of recycling and doping LiMn1/3Ni1/3Co1/3O2 of lithium-ion battery with dimethyl sulfoxide (DMSO) instead of N-methylpyrrolidone (NMP) on the electrochemical performance of the battery has been investigated for the first time. Observation shows that preparing the cathode active materials with dimethyl sulfoxide will increase the conductivity of the battery. The results show that the as-recovered LiMn1/3Ni1/3Co1/3O2 modified with LiOH · H2O calcined at 450°C delivers discharge capacities of about 247 mA h g−1 in the first cycle with discharge efficiency of 83.1% in sample doped with dimethyl sulfoxide, and 189 mA h g−1 with discharge efficiency of 82.7% in N-methylpyrrolidone at the rate of 0.2 C. The asrecovered samples calcined at 800 and 850°C deliver 149 and 217 mA h g−1 in the fourth cycles respectively in dimethyl sulfoxide. The capacity loss observed in dimethyl sulfoxide faded with increase in cycle numbers. In general, for the samples doped with dimethyl sulfoxide, better performances were evident with high discharge capacities in powders calcined at a lower temperature than higher temperature in accordance with particle sizes shown by the SEM images. On the basis of better cyclic performance of lithium metal cathode and environmental safety, it is evident that relatively cheap dimethyl sulfoxide could replace N-methylpyrrolidone in battery formulations. The X-ray diffraction patterns revealed that LiMn1/3Ni1/3Co1/3O2 was successfully recycled by dimethyl sulfoxide.
Similar content being viewed by others
References
Spillman, D.M. and Takeuchi, E.S., US Patent 5935728, 1999.
Tackeuchi, E.S. and Walsh, K.M., US Patent 5614331, 1997.
Ó’Laoire, C.M., Investigations of oxygen reduction reactions in non-aqueous electrolytes and the lithiumair battery, A PhD Chemistry Dissertations, Department of Chemistry and Chemical Biology, Boston, Massachusetts: Northeastern University, 2010.
Chua, D., Develop a Low Cost, Safe and Environmentally Benign High Energy and High Rate Reserve Battery SERDP Project Number: 1360 MaxPower, Inc., 2004.
Microelectronic Applications using DMSO (Dimethyl Sulfoxide). Gaylord Chemical Company, L.L.C., 2008, www.gaylordchcmical.com.
Nogami, T. and Nawa, M., US Patent 4 510 216, 1985.
Filler, R., Luo, K., and Mandal, B., Solid State Ionics, 2006, vol. 177, pp. 857–861.
Coustier, F., Battery Cell Separator and Fabrication Process, WO 2002050929, 2002, www.gaylordchcmical.com.
Morita, M., Tachihara, F., and Matsuda, Y., Electrochimica Acta, 1987, vol. 32, pp. 299–305.
Vignes, R.P., A superior solvent underutilized because of a safety myth, in Dimethyl Sulfoxide (DMSO), VA: Semiconductor Safety Association Annual Meeting Arlington, April 25–28, 2000.
Okuno, H., Koshina, H., and Morita, A., Patent 5525443, 1996.
Chang, Z.R., Liu, Y., and Tang, H.W., Electrochem. Solid-State Lett., 2011, vol. 14, pp. A90–A92.
Liu, S.S., Yang, J., and Wang, F., Acta Chimica Sinica, 2009, vol. 67, pp. 2395–2401.
Li, J., Zhang, Q., and He, X., Met. Soc. China, 2007, vol. 17, pp. s897–s901.
Belharouak, I., Sun, Y.-K., and Liu, J., J. Power Sources, 2003, vol. 123, pp. 247–252.
Oh, S.W., Park, S.H., and Park, C.-W., Solid State Ionics, 2004, vol. 171, pp. 167–172.
Wu, F., Wang, M., and Su, Y., J. Power Sources, 2010, vol. 195, pp. 2362–2367.
Gangulibabu, N., Bhuvaneswari, D., and Kalaiselvi, N., J. Solid State Electrochem., 2013, vol. 17, pp. 9–17.
Sclar, H., Kovacheva, D., and Zhecheva, E., J. Electrochem. Soc., 2009, vol. 156, pp. A938–A948.
Iqbal, M.Z., Ali, S., and Mirza, M.A., CODEN JNSMAC, 2008, vol. 48, pp. 51–63.
Dubarry, M. and Liaw, B.Y., J. Power Sources, 2009, vol. 194, pp. 541–549.
Koltypin, M., Aurbach, D., Nazar, L., and Ellis, B., J. Power Sources, 2007, vol. 174, pp. 1241–1250.
Striebel, K.A., Guerfi, A., Shim, J., Armand, M., Gauthier, M., and Zaghib, K., J. Power Sources, 2003, vols. 119–121, p. 951.
Striebel, K., Shim, J., Sierra, A., Yang, H., Song, X., Kostecki, R., and McCarthy, K., J. Power Sources, 2005, vol. 146, p. 33.
Zhang, Y., Wang, C.-Y., and Tang, X., J. Power Sources, 2011, vol. 196, pp. 1513–1520.
Aurbach, D., Gamolsky, K., Markovsky, B., Salitra, G., and Gofer, Y., J. Electrochem. Soc., 2000, vol. 147, p. 1322.
Ding, C.X., Meng, Q.S., and Wang, L., Materials Research Bulletin, 2009, vol. 44, pp. 492–498.
Ren, H.B., Huang, Y.H., Wang, Y.H., Li, Z., Cai, P., Peng, Z., and Zhou, Y., Mater. Chem. Phys., 2009, vol. 117, pp. 41–45.
Cheralathan, K.K., Kang, N.Y., Park, H.S., Lee, Y.J., Choi, W.C., Ko, Y.S., and Park, Y.-K., J. Power Sources, 2010, vol. 195, pp. 1486–1494.
MTI Corporation, N-methylpyrrolidinone; N-methyl-2-pyrrolidone; MSDS, 2009.
Baker, J.T., NMP Environmental Health and Safety, MSDS Number: M7114, 2009.
Baker, J.T., N,N-dimethyl acetamide, in Environmental Health and Safety, MSDS Number: D5784, 2008.
EMD Chemicals Inc., N,N-dimethylformamide, biosynthesis, for peptide synthesis, 2003.
Baker, J.T., Dimethyl sulfoxide, Environmental Health and Safety, Material Safety Data Sheet, MSDS Number: D7120, 2008.
ChemicalLAND21.com 2000–2008.
Massachusetts Chemical Fact Sheet, The Massachusetts Toxics Use Reduction Institute, pp. 1–6, http://www.turi.org/.
Tolando, R., Zanovello, A., and Ferrara, R., Toxicology Lett., 2001, vol. 124, pp. 101–111.
Baker, J.T., N,N-dimethylformamide, Environmental Health and Safety, Material Safety Data Sheet, MSDS Number: D6408, 2009.
Taminco, N-methylpyrrolidone electronic grade, Technical Data Sheet, TDS 51124/03-2011, 1–4, 2011, www.taminco.com.
Taminco, People and molecules, Dimethylacetamide, Technical Data Sheet 51093/10-2010, 1–4, 2010, pp. 1–4, www.taminco.com.
Taminco, Dimethyl formamide, Technical Data Sheet TDS51021/0204, 2004, www.taminco.com.
Dimethyl Sulfoxide (DMSO) Physical Properties, Gaylord Chemical Corporation, 2005, pp. 1–16, www.gaylordchemicai.com.
Sigma-Aldrich, Sigma-Aldrich Corporation, 2011, http://www.sigmaaldrich.com.
Japan, Chemicals Evaluation and Research Institute (CERI), Hazard Assessment Report on N,N-Dimethyl formamide, CAS no. 68-12-2, 2007.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Russian in Elektrokhimiya, 2014, Vol. 50, No. 9, pp. 991–998.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Bankole, O.E., Gong, C. & Lei, L. Environmentally compatible dimethyl sulfoxide: an alternative to N-methylpyrrolidone for electrochemical performance of recycled LiMn1/3Ni1/3Co1/3O2 in lithium-ion battery. Russ J Electrochem 50, 891–898 (2014). https://doi.org/10.1134/S1023193514030021
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193514030021