Abstract
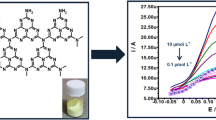
In this paper, a novel poly(aminosulfonic acid) modified glassy carbon electrode (PASA/GCE) for the determination of Sudan II was fabricated through electrochemical polymerizat ion. The electrochemical behavior of Sudan II at the modified electrode was studied by cyclic voltammetry. Results show that the modified electrode exhibits excellent electrocatalytic activity toward the electrochemical redox reaction of Sudan II. Under optimal experimental conditions, the oxidation peak current is linearly proportional to the concentration of Sudan II in the ranges of 4.0 × 10−8 to 1.0 × 10−6 mol L−1 and 1.0 × 10−6 to 1.2 × 10−5 mol L−1. The linear regression equations are i pa(A) = 2.87c + 3.74 × 10−6, r = 0.9977 and i pa(A) = 0.78c + 6.11 × 10−6, r = 0.9982, respectively, and the detection limit is 4.0 × 10−9 mol L−1. The novel method shows good recovery, reproducibility and sensitivity for the voltammetric determination of Sudan II in food samples.
Similar content being viewed by others
References
Ruf, J., Walter, P., Kandler, H., and Kaufmann, A., Food Additives & Contaminants: Part A, 2012, vol. 29, p. 897.
Ahmed Refat, N.A.G., Ibrahim, Z.S., Moustafa, G.G., Sakamoto, K.Q., Ishizuka, M., and Fujita, S., J. Biochem. Mol. Toxicol., 2008, vol. 22, p. 77.
Xu, H., Heinze, T.M., Chen, S., Cerniglia, C.E., and Chen, H., Applied and Environmental Microbiology, 2007, vol. 73, p. 7759.
Mao, J., Wang, J., Meng, J., and Zhao, S., Chinese Journal of Analysis Laboratory, 2009, vol. 28, p. 341.
Rapid Alert System for Food and Feed, Available at http://ec.europa.eu/food/food/rapidalert/index-en.htm, vol. (accessed 17.06.2012)
Erta, E., Özer, H., and Alasalvar, C., Food Chem., 2007, vol. 105, p. 756.
Zhang, Y.P., Zhang, Y.J., Gong, W.J., Gopalan, A.I., and Lee, K.-P., J. Chromatogr. A, 2005, vol. 1098, p. 183.
Mazzotti, F., Di Donna, L., Maiuolo, L., Napoli, A., Salerno, R., Sajjad, A., and Sindona, G., J. Agric. Food. Chem., 2007, vol. 56, p. 63.
Murty, M.R.V.S., Sridhara Chary, N., Prabhakar, S., Prasada Raju, N., and Vairamani, M., Food Chem., 2009, vol. 115, p. 1556.
Cornet, V., Govaert, Y., Moens, G., Van Loco, J., and Degroodt, J.-M., J. Agric. Food. Chem., 2006, vol. 54, p. 639.
Zhang, Y., Zhang, Z., and Sun, Y., J. Chromatogr. A, 2006, vol. 1129, p. 34.
Fan, Y., Chen, M., Shentu, C., El-Sepai, F., Wang, K., Zhu, Y., and Ye, M., Anal. Chim. Acta, 2009, vol. 650, p. 65.
Li, C., Yang, T., Zhang, Y., and Wu, Y.-L., Chromatographia, 2009, vol. 70, p. 319.
Wu, L.P., Li, Y.F., Huang, C.Z., and Zhang, Q., Anal. Chem., 2006, vol. 78, p. 5570.
Ensafi, A.A., Rezaei, B., Amini, M., and Heydari-Bafrooei, E., Talanta, 2012, vol. 88, p. 244.
Lijun, L., Laibo, Y., Hao, C., Qifeng, C., Fengmin, W., Tian, C., Xiaoyong, Z., Hongxing, K., and Jianling, W., Anal Lett., 2007, vol. 40, p. 3290.
Yang, B., Hu, F., Wei, J., and Wang, C., Acta Chim. Sinica, 2009, vol. 67, p. 2585.
Lin, X., Kang, G., and Lu, L., Bioelectrochemistry, 2007, vol. 70, p. 235.
Deinhammer, R.S., Ho, M., Anderegg, J.W., and Porter, M.D., Langmuir, 1994, vol. 10, p. 1306.
Santos, D.P., Zanoni, M.V.B., Bergamini, M.F., Chiorcea-Paquim, A.-M., Diculescu, V.C., and Oliveira Brett, A.-M., Electrochim. Acta, 2008, vol. 53, p. 3991.
Bard, A.J. and Faulkner, L.R., Electrochemical Methods, Fundamentals and Applications, New York: John WileyI & Sons, Inc., 2001.
Yang, D., Zhu, L., and Jiang, X., J. Electroanal. Chem., 2010, vol. 640, p. 17.
Yuan, J., Liao, L., Lin, Y., Deng, C., and He, B., Anal. Chim. Acta, 2008, vol. 607, p. 160.
Yang, D., Zhu, L., Jiang, X., and Guo, L., Sens. Actuators, B, 2009, vol. 141, p. 124.
Yin, H., Zhou, Y., Meng, X., Tang, T., Ai, S., and Zhu, L., Food Chem., 2011, vol. 127, p. 1348.
Mo, Z., Zhang, Y., Zhao, F., Xiao, F., Guo, G., and Zeng, B., Food Chem., 2010, vol. 121, p. 233.
Laviron, E., J. Electroanal Chem. Interfacial Electrochem., 1979, vol. 101, p. 19.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Elektrokhimiya, 2013, Vol. 49, No. 11, pp. 1179–1187.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Ma, X., Chao, M. Sensitive electrochemical determination of Sudan II in food samples using a poly(aminosulfonic acid) modified glassy carbon electrode. Russ J Electrochem 49, 1057–1064 (2013). https://doi.org/10.1134/S1023193513110086
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193513110086