Abstract
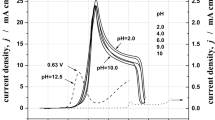
The regularities of electrochemical deposition and dissolution of gold in the mixed sulfite-thiocarbamide electrolytes in the absence and in the presence of sodium sulfide additive are studied by using the voltammetric measurements on a renewable electrode and quarts microgravimetry. It is shown that, in the cathodic metal deposition, an addition of sodium sulfide promotes the depolarization effect, which is caused by the presence of thiocarbamide in the solution. Under the anodic polarization of gold in the mixed sulfite-thiocarbamide solution with pH < 10, the gold dissolution rate is insignificant. An addition of 10−5 M Na2S to this solution dramatically accelerates the process. At pH > 10, the gold dissolution in the sulfite-thiocarbamide electrolyte is observed even in the solution free of Na2S additive. It is evidenced that this is associated with spontaneous accumulation of sulfide-containing species in the solution, probably, as a result of thiocarbamide hydrolysis; the rate of hydrolysis steeply increases with increasing pH value.
Similar content being viewed by others
References
Horkans, J. and Romankiw, L.T., J. Electrochem. Soc., 1977, vol. 124, p. 1499.
Bor, P., Tomkyavichyus, G.A., Butkyavichyus, Yu.P., Yuodkazis, K.I., Srb, M., and Vrobel, L., Tr. Akad. Nauk Lit. SSR, Ser. B., 1981, vol. 2, p. 123.
Bek, R.Yu. and Lavrova, T.A., Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk, 1971, no. 4, p. 17.
Bek, R.Yu., Zh. Prikl. Khim., 1997, vol. 70, p. 420.
Shevtsova, O.N., Bek, R.Yu., Zelinskii, A.G., and Vais, A.A., Russ. J. Electrochem., 2006, vol. 42, p. 239.
Chai, L., Okido, M., and Wei, W., Hydrometallurgy, 1999, vol. 53, p. 255.
Wie, D., Chai, L., Ichino, R., and Okido, M., J. Electrochem. Soc., 1999, vol. 146, p. 559.
Shevtsova, O.N., Zelinskii, A.G., and Bek, R.Yu., Russ. J. Electrochem., 2009, vol. 45, p. 818.
Bek, R.Yu. and Shevtsova, O.N., Russ. J. Electrochem., 2011, vol. 47, p. 1029.
Bek, R.Yu., Shevtsova, O.N., and Zelinskii, A.G., Russ. J. Electrochem., 2012, vol. 48, p. 931.
Shaw, H.R. and Walker, D.G., J. Am. Chem. Soc., 1956, vol. 78, no. 22, p. 5769.
Zelinskii, A.G. and Bek, R.Yu., Elektrokhimiya, 1985, vol. 21, p. 66.
Zhang, S. and Nicol, M.J., J. Appl. Electrochem., 2003, vol. 33, p. 767.
Jeffrey, M.J., Hydrometallurgy, 2001, vol. 60, p. 7.
Aleksandrova, T.P., Ovchinnikova, S.N., Vais, A.A., and Bek, R.Yu., Zh. Anal. Khim., 1999, vol. 54, p. 732.
Sauerbrey, G., Z. Phys., 1959, vol. 155, p. 206.
Vatankhah, G., Lessard, J., Jerkiewicz, G., Zolfaghari, A., and Conway, B.E., Electrochim. Acta, 2003, vol. 48, p. 1613.
Bek, R.Yu. and Rogozhnikov, N.A., Russ. J. Electrochem., 1997, vol. 33, p. 579.
Bek R.Yu., Shevtsova O.N., and Shuraeva L.I, Rus. J. Electrochem., 2008, vol. 44, p. 1263.
Belevantsev, V.N., Peshchevitskii, B.N., and Tsvelodub, L.D., Zh. Neorg. Khim., 1986, vol. 31, p. 3065.
Peshchevitskii, B.I. and Erenburg, A.M., Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk, 1970, no. 4, p. 83.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © R.Yu. Bek, O.N. Novgorodtseva-Shevtsova, 2013, published in Elektrokhimiya, 2013, Vol. 49, No. 5, pp. 530–535.
Rights and permissions
About this article
Cite this article
Bek, R.Y., Novgorodtseva-Shevtsova, O.N. The role of sulfide ions in the electrode processes involving gold in sulfite-thiocarbamide electrolytes. Russ J Electrochem 49, 470–474 (2013). https://doi.org/10.1134/S1023193513050029
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193513050029