Abstract
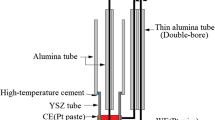
The Tubandt method of electrolysis is used for studying the nature of ionic carriers in ceramics of tungstates Me2+{WO4} (Ca, Sr, Ba) and Me 3+2 {WO4}3 (Al, Sc, In) which are solid electrolytes. These compounds have the salt-like islet structure with isolated tetrahedrons {WO 2−4 } and are crystallized in the allied structural types of scheelite (CaWO4) for Me2+ and Sc tungstate (Sc2{WO4}3) for Me3+. The electrolysis is carried out in 2- or 3-section cells (−)Pt|M n/n+2 {WO4}|Me n/n+2 {WO4}|Pt(+) in air atmosphere at the temperature of ∼900°C and cell voltage of 4 and 300 V. All experiments without exception demonstrate a decrease in the mass of the cathodic section of cells. This points to the negative charge of ionic mass carriers and their transfer towards the Pt(+) electrode. The cathodic briquette mass loss Δm (−) depends linearly on the charge passed through a cell. In all experiments with MeWO4 tungstates, the anodic disk mass remains constant. The electrolysis of Me2(WO4)3 cells is always accompanied by an increase in the anolyte mass Δm (+); however, in all experiments, Δm (−) > Δm (+). All data on mass variation and the results of studying the composition of nearelectrode electrolyte layers by XRD and SEM methods correspond to the condition \(t_{WO_4^{2 - } } > t_C \) (C is the cation), i.e., {WO 2−4 } anions pertain to the major ionic carriers. The transport number \(t_{WO_4^{2 - } } \) is calculated based on the Faraday law from Δm (−). It is shown that the second ionic carrier with the mobility even higher than that of {WO 2−4 } is the O2− ion. For middle values of transport numbers, their ratio is shown to be \(t_{O^{2 - } } \)(0.5–0.8) > (0.2–0.5)\(t_{WO_4^{2 - } } \). No results that would confirm the involvement of Me2+ and Me3+ ions in conduction are obtained.
Similar content being viewed by others
References
Jander, W.Z., Inorg. Chem., 1927, vol. 163, nos. 1–2, p. 1.
Jander, W.Z., Inorg. Chem., 1928, vol. 174, nos. 1–2, p. 11.
Gurvich, A.M., Rentgenolyuminofory i rentgenovskie ekrany (X-ray Phosphors and X-ray Screens), Moscow: Atomizdat, 1976.
Kaminskii, A.A., Lazernye kristally (Laser Crystals), Moscow: Nauka, 1975.
Vashuk, V.V. and Kononyuk, I.F., Dokl. Akad. Nauk BSSR, 1975, vol. 19, no. 12, p. 1098.
Zhukovskii, V.M., Doctoral (Chem.) Dissertation, Sverdlovsk: Ural Politekhn. In-t im. S.M. Kirova, 1974.
Fedorova, L.M., Cand. Sci. (Chem.) Dissertation, Sverdlovsk: Ural. Gos. Un-t, 1983.
Zhukovskii, V.M., Tkachenko, E.V., and Zhukovskaya, A.S., in Fizika metallov i ikh soedinenii (Physics of Metals and their Compounds), Sverdlovsk: UPI, 1976, p. 49.
Tkachenko, E.V., Zhukovskii, V.M., Neiman, A.Ya., et al., Dokl. Akad. Nauk SSSR, 1977, vol. 233, no. 6, p. 1106.
Neiman, A.Ya., Solid State Ionics, 1996, vol. 83, nos. 3–4, p. 263.
Neiman, A.Ya., Guseva, A.F., and Kalyakin, A.S., Izv. Akad. Nauk SSSR, Neorg. Mater., 1990, vol. 26, no. 8, p. 1728.
Neiman, A.Ya., Doctoral (Chem.) Dissertation, Sverdlovsk: In-t Vysokotemper. Elektrokhimii UNTs AN SSSR, 1989.
Imanaka, N. and Tamura, S., Bull. Chem. Soc. Jpn., 2011, vol. 84, no. 4, p. 353.
Driscoll, D., Islam, S., and Slater, P., Solid State Ionics, 2005, vol., nos. 5–6, p. 176.
Adams, S., Rao, P., Edwards, D., Neiman, A., and Pestereva, N., Chemistry of Materials, 2008, vol. 20, no. 20, p. 6335.
Zhou, K. and Adams, S., Chem. Monthly, 2009, vol. 140, no. 9, p. 1017.
Zhou, Y., Neiman, A., and Adams, S., Physica Status Solidi B, vol. 248, no. 1, p. 130.
Zhou, Y., Rao, P., and Adams, S., Solid State Ionics, 2011, vol. 192, no. 1, p. 34.
Kulikova, T., Neiman, A., Kartavtseva, A., et al., Solid State Ionics, 2008, vol. 178, nos. 33–34, p. 1714.
The Handbook of Solid State Electrochemistry, Gellings, P.J. and Bouwmeester, H., Eds., CRC-PRESS, 1997.
Diagrammy sostoyaniya tugoplavkikh oksidov: Sprav. Vyp. 5. Dvoinye sistemy (Phase Diagrams of Refractory Oxides, Handbook, No. 5, Binary Systems), Leningrad: Nauka, 1988, ch. 4, p. 348.
Zelikman, A. and Korshunov, B., Metallurgiya redkikh metallov (Metallurgy of Rare Metals), Moscow: Metallurgiya, 1991.
Ziolkowski, J. and Dziembai, L., J. Solid State Chem., 1985, vol. 57, no. 3, p. 291.
Brown, I.D. and Shannon, R.D., Acta Cristallogr. A, 1973, vol. 29, no. 3, p. 266.
Evans, R., Vvedenie v kristallokhimiyu (Introduction to Crystallochemistry), Moscow: Goskhimizdat, 1948.
Pestereva, N.N., Sharafutdinov, A.R., and Kostikov, Yu.P., Russ. J. Electrochem., 2005, vol. 41, p. 598.
Pestereva, N.N., Zhukova, A.Yu., and Neiman, A.Ya., Russ. J. Electrochem., 2007, vol. 43, p. 1305.
Kazenas, E.K. and Tsvetkov, Yu.V., Isparenie oksidov (Evaporation of Oxides), Moscow: Nauka, 1997.
Knozinger, H. and Taglauer, E., Catalysis. Royal Society of Chemistry, 1993, vol. 10, p. 40.
Ukshe, E.A., Ukshe, A.E., and Bukun, N.G., in Issledovaniya v oblasti khimii ionnykh rasplavov i tverdykh elektrolitov. Sb. nauch. tr (Studies in the Field of Chemistry of Ionic Melts and Solid Electrolytes, Collection of Papers), Kiev: Naukova Dumka, 1985, p. 3.
Neiman, A.Ya. and Guseva, A.F., Russ. J. Electrochem., 1993, vol. 29, p. 1386.
Neiman, A.Ya., Rus. J. Phys. Chem., 2001, vol. 75, no. 12, p. 1945.
Kazenas, E.K., Termodinamika ispareniya dvoinykh oksidov (Thermodynamics of Evaporation of Binary Oxides), Moscow: Nauka, 2004.
Sawada, S., Ando, R., and Nomura, S. Phys. Rev., 1951, vol. 84, no. 5, p. 1054.
Karapetyan, A., Pestereva, N., and Neiman, A., Abstracts of Papers Solid State Chemistry-2010, Demel, J. and Grygar, T., Eds., Prague, 2010, p. 208.
Porai-Koshits, M.A. and Atovmyan, L.O., in Itogi Nauki Tekh., Ser.: Kristallokhim., Moscow: VINITI, 1985, no. 19, p. 3.
Andersen, N.H., et al., Solid State Ionics, 1992, vol. 57, nos. 3–4, p. 203.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.Ya. Neiman, N.N. Pestereva, Y. Zhou, D.O. Nechaev, E.A. Koteneva, K. Vanec, B. Higgins, N.A. Volkova, I.G. Korchuganova, 2013, published in Elektrokhimiya, 2013, Vol. 49, No. 9, pp. 999–1012.
Rights and permissions
About this article
Cite this article
Neiman, A.Y., Pestereva, N.N., Zhou, Y. et al. The nature and the mechanism of ion transfer in tungstates Me2+{WO4} (Ca, Sr, Ba) and Me 3+2 {WO4}3 (Al, Sc, In) according to the data acquired by the tubandt method. Russ J Electrochem 49, 895–907 (2013). https://doi.org/10.1134/S1023193512120075
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193512120075
Keywords
- tungstates with the scheelite and Sc2(WO4)3 structures
- co-anion conduction type
- determination of transport numbers for WO 2−4 anions by the Tubandt method
- catholyte mass loss
- electrolysis products
- the absence of charge transfer by Me2+ and Me3+ cations
- models of cooperative nonreconstructive migration of WO 2−4