Abstract
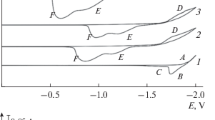
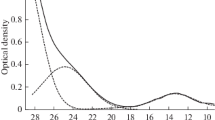
The process of dissolution of titanium, silicon, and boron oxides in carbonate melts is studied. Voltammetric studies are used to show that the most electropositive electrochemical process in molten carbonates is discharge of carbon dioxide dissolved in the melt to elementary carbon. Electrochemical synthesis of carbides in carbonate melts is possible at the potentials more positive by their value than the potential of carbonate decomposition. Interaction of titanium and silicon carbides with carbonate solutions is studied.
Similar content being viewed by others
References
Samsonov, G.V. and Vinitskii, I.M., Tugoplavkie soedineniya (Refractory Compounds), Moscow: Metallurgiya, 1976.
Gurin, V.N., Usp. Khim., 1972, vol. 41, p. 616.
Material Science and Technology. Structure and Properties of Ceramics, Swain, M.V., ed., Weinheim: VCH, 1994, vol. 11.
Delimarskii, Yu.K., Grishchenko, V.F., Tumanova, N.Kh., and Shapoval, V.I., Ukr. Khim. Zh., 1970, vol. 36, no. 2, p. 136.
Ingram, M.D., Baron, B., and Janz, G.J., Electrochim. Acta, 1966, vol. 11, no. 11, p. 1629.
Elwell, O., Mater. Res. Bull., 1982, vol. 17, no. 6, p. 697.
Diagrammy plavkosti solevykh sistem. Ch. 2 (Fusibility Diagrams of Salt Systems. Part 2), Posypaiko, V.P., ed., Moscow: Metallurgiya, 1977.
Nekrasov, B.V., Osnovy obshchei khimii (Fundamentals of General Chemistry), Moscow: Khimiya, 1969, vol. 2.
Jacobson, N.S., J. Am. Ceram. Soc., 1986, vol. 69, p. 74.
Liu, G., Li, M., Yanchun, Zhou., and Zhang, Y., J. Eur. Ceram. Soc., 2003, vol. 23, p. 1957.
Barin, I., Thermodynamic data of pure substances, Weinheim: VCH, 1993.
Nicholson, R.S. and Shain, I., Anal. Chem., 1964, vol. 36, p. 706.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © S.V. Devyatkin, 2012, published in Elektrokhimiya, 2012, Vol. 48, No. 10, pp. 1105–1109.
Rights and permissions
About this article
Cite this article
Devyatkin, S.V. Chemical and electrochemical behavior of titanium, silicon, and boron oxides in molten carbonates. Russ J Electrochem 48, 1000–1004 (2012). https://doi.org/10.1134/S1023193512100072
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193512100072