Abstract
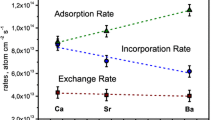
The oxygen exchange and diffusion kinetics are studied by means of isotopic exchange in complex oxides La0.80Sr0.20Ga0.85 − x Mg0.15Co x O3 − δ (x = 0.00, 0.05, 0.15, 0.20, 0.25) and in electrolyte La0.88Sr0.12Ga0.82Mg0.18O3 − δ with gold-coated surface. The oxygen exchange rates and diffusion coefficients are determined in the bulk material and near the surface within the range of temperatures from 600 to 900°C and oxygen pressures from 0.3 to 1.0 kPa. The activation energies of the oxygen exchange and diffusion processes are calculated. By cobalt doping, gallium sublattice is shown to increase the interphase exchange rate; however, the oxygen transport properties in the bulk phase are not significantly affected by cobalt, while cobalt additive decreases the activation barrier of oxygen diffusion in the near-surface area. Gold activates the electrolyte surface, affecting the exchange rate and increasing the first-type exchange share. Cobalt substitution increases the third-type exchange share as compared to that of La0.88Sr0.12Ga0.82Mg0.18O3 − δ (x = 0.00, 0.05, 0.15, 0.20, 0.25) and in elec. The oxygen exchange with gas phase is assumed to be limited by molecular oxygen adsorption-desorption process on the electrolyte surface.
Similar content being viewed by others
References
Kharton, V.V., Shaula, A.L., Vyshatko, N.P., and Margues, F.M.B., Electrochim. Acta, 2003, vol. 48, p. 1817.
Trofimenko, N. and Ullmann, H., Solid State Ionics, 1999, vol. 124, p. 263.
Khorkounov, B.A., Nafe, H., and Aldinger, F., Solid State Electrochem., 2006, vol. 10, p. 479.
Thangadurai, V. and Weppner, W., Electrochim. Acta, 2005, vol. 50, p. 1871.
Shkerin, S.N., Bronin, D.I., Kovyazina, S.A., Gorelov, V.P., Kuzmin, A.V., Martemyanova, Z.S., and Beresnev, S.M., Solid State Ionics, 2004, vol. 171, p. 129.
Slater, P.R., Irvine, J.T.S., Ishihara, T., and Takita, Y., J. Solid State Chem., 1998, vol. 139, p. 139.
Ishihara, T., Akbay, T., Furutani, H., and Takita, Y., Solid State Ionics, 1998, vols. 113–115, p. 585.
Polini, R., Falsetti, A., Traversa, E., Schaf, O., and Knauth, Ph., J. European Ceramic Soc., 2007, vol. 27, p. 4291.
Yamaji, K., Horita, T., Sakai, N., and Yokokawa, H., Solid State Ionics, 2002, vols. 152–153, p. 517.
Vdovin, G.K., Kurumchin, E.Kh., Isaeva, E.V., and Bronin, D.I., Elektrokhimiya, 2001, vol. 37, p. 347 [Russ. J. Electrochem. (Engl. Transl.), vol. 37, p.].
Klier, K. and Kucera, E., J. Physics Chem. Solids, 1966, vol. 27, p. 1087.
Ezin, A.N., Kurumchin, E.Kh., Murygin, I.V., Tsidilkovski, V.I., and Vdovin, G.K., Solid State Ionics, 1998, vol. 112, p. 117.
Muzykantov, V.S., Panov, G.I., and Boreskov, G.K., Kinetika i Kataliz, 1973, vol. 14, p. 948.
Ezin, A.N., Tsidilkovski, V.I., and Kurumchin, E.Kh., Solid State Ionics, 1996, vol. 84, p. 105.
Boreskov, G.K., Geterogennyi kataliz (Heterogeneous Catalysis), Moscow: Nauka, 1986.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.Kh. Kurumchin, M.V. Ananjev, G.K. Vdovin, M.G. Surkova, 2010, published in Elektrokhimiya, 2010, Vol. 46, No. 2, pp. 213–220.
Rights and permissions
About this article
Cite this article
Kurumchin, E.K., Ananjev, M.V., Vdovin, G.K. et al. Exchange kinetics and diffusion of oxygen in systems based on lanthanum gallate. Russ J Electrochem 46, 205–211 (2010). https://doi.org/10.1134/S1023193510020126
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193510020126