Abstract
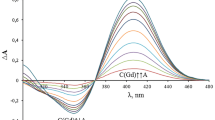
The Au(III)-luteolin system was studied by means of cyclic voltammetry, spectrometry, and quantum chemical simulation. The mutual effect of luteolin to Au(III) reduction and Au(III) to luteolin oxidation was studied by means of cyclic voltammetry on Pt and carbon glass electrodes in 0.05 M tris-buffer solution (pH 8) containing ethyl alcohol. The absorption spectra of luteolin were recorded with and without Au(III) in 0.05 M tris-buffer solution (pH 8) containing ethyl alcohol. The quantum chemical simulation of Au(III)-tris, Au(III)-luteolin, and Au(III)-tris-luteolin systems was carried out. On the basis of the collected data, formation of Au(III)-tris-luteolin complex in 0.05 M tris-buffer solution (pH 8) in the presence of ethanol was suggested.
Similar content being viewed by others
References
Levchenko, L.A., Lariontseva, N.V., and Sadkov, A.P., Dokl. Akad. Nauk, 1993, vol. 333, p. 392.
Levchenko, L.A., Sadkov, A.P., Lariontseva, N.V., Kulikova, V.S., Shilova, A.K., and Shilov, A.E., Dokl. Akad. Nauk, 2004, vol. 294, p. 272 [Dokl. (Engl. Transl.), vol. 294, p.].
Kulakovskaya, S.I., Levchenko, L.A., Kulikov, A.V., Sadkov, A.P., Shestakov, A.F., and Pichugina, D.A., Elektrokhimiya, 2007, vol. 43, p. 94 [Russ. J. Electrochem. (Engl. Transl.), vol. 43, p.].
Levchenko, L.A., Kartsev, V.G., Sadkov, A.P., Shestakov, A.F., Shilova, A.K., and Shilov, A.E., Dokl. Akad. Nauk, 2007, vol. 412, p. 500 [Dokl. (Engl. Transl.), vol. 412, p.].
Zoulis, N.E. and Efstathiou, C.E., Anal. Chim. Acta, 1996, vol. 320, p. 255.
Volikakis, G.J. and Efstathiou, C.E., Talanta, 2000, vol. 51, p. 775.
Van Acker, S.A.B.E., De Groot, M.J., Berg, D.J., Tromp, M.N.J.L., Kelder, G., Donne-Op, Van Der Vijgh W.J.F., and Bast, A., Chem. Res. Toxicol., 1996, vol. 9, p. 1305.
Hendrickson, H.P., Kaufman, A.D., and Lunte, C.E., J. Pharm. Biomed. Anal, 1994, vol. 12, p. 325.
Hendrickson, H.P., Sahafayen, M., Bell, M.A., Kaufman, A.D., Hadwiger, M.E., and Lunte, C.E., J. Pharm. Biomed. Anal, 1994, vol. 12, p. 335.
Yacynych, A.M. and Mark, H.B., J. Electrochem. Soc., 1976, vol. 123, p. 1346.
Adkins, H. and Cox, F.W., J. Am. Chem. Soc., 1938, vol. 60, p. 1151.
Engelkemeier, D.W., Geissman, T.A., Crowell, W.R., and Friess, S.L., J. Am. Chem. Soc., 1947, vol. 69, p. 155.
Geissman, T.A. and Friess, S.L., J. Am. Chem. Soc., 1949, vol. 71, p. 3893.
Gottlieb, O.R., The Flavonoids. Part 1, Harbone, J.B., Mabry, T.J., and Marby, H., Eds., New York: Academic, 1975, p. 311.
Hodnick, W.F., Milosavljevic, E.B., Nelson, J.H., and Pardini, R.S., Biochem. Pharmac., 1988, vol. 37, p. 2607.
Nicholson, R.S. and Shain, J., Anal. Chem., 1964, vol. 36, p. 706.
Cotton, F.A. and Wilkinson, G., Advanced Inorganic Chemistry, New York: Wiley Eastern, 1972, p. 1044.
Paddefet, R., in Khimiya zolota (Chemistry of Gold), Moscow: Mir, 1982.
Rich, R.L. and Taube, H., J. Phys. Chem., 1954, vol. 58, p. 6.
Dubinskii, V.I., Shul’man, V.M., and Peshchevitskii, B.I., Zh. Neorg. Khim., 1968, vol. 13, p. 54.
Carlsson, L. and Lundgren, G., Acta Chem. Scand., 1967, vol. 21, p. 819.
Peshchevitskii, B.I., Belevantsev, V.I., and Kurbatova, N.V., Zh. Neorg. Khim., 1971, vol. 16, p. 1898.
Skibsted, L.H. and Bjerrum, J., Acta Chem. Scand. Ser. A, 1974, vol. 28, p. 740.
Weishaupt, M. and Straehle, J., Naturforsch. B, 1976, vol. 31B, p. 554.
Block, B.P. and Bailar, J.K., J. Am. Chem. Soc., 1951, vol. 73, p. 4722.
Kharasch, M.S. and Isbell, H., J. Am. Chem. Soc., 1931, vol. 53, p. 3059.
Gibson, C.S. and Colles, W.M., J. Chem. Soc., 1931, p. 2407.
Monoghan, P.K. and Puddephatt, R.J., Inorg. C him. Acta, 1975, vol. 15, p. 231.
Dell’Amico, D.B. and Calderazzo, F., Gazz. Chim. Ital., 1973, vol. 103, p. 1099.
Calderazzo, F. and Dell’Amico, D.B., J. Organomet. Chem., 1974, vol. 76, p. C59.
Huttel, R. and Konietzny, A., Chem. Ber., 1973, vol. 106, p. 2098.
Dubois, R.J., Hagimassy, J., Noble, A.C., and Popp, F.D., J. Heterocycl. Chem., 1966, vol. 3, p. 377.
Harris, C.M., J. Chem. Soc., 1959, p. 682.
Harris, C.M. and Lockyer, T.N., J. Chem. Soc., 1959, p. 3083.
Mundorf, T. and Dehnicke, K., Z. Anorg. Allg. Chem., 1974, vol. 408, p. 146.
Charlton, R.J., Harris, C.M., Patil, H., and Stephenson, N.G., Inorg. Nucl. Chem. Lett., 1966, vol. 2, p. 409.
Robinson, W.T. and Sinn, E., J. Chem. Soc., Dalton Trans., 1975, p. 726.
Fleischer, E. and Laszlo, A., Inorg. Nucl. Chem. Lett., 1969, vol. 5, p. 373.
Timkovich, R. and Tulinsky, A., Inorg. Chem., 1977, p. 962.
Chia, P.S.K. and Livingstone, S.E., Aust. J. Chem., 1968, vol. 21, p. 339.
Barclay, G.A., Harris, C.M., and Kingston, J.V., Chem. Ind., 1965, p. 227.
Willigen, H. and van Rens, J.G.M., Chem. Phys. Lett., 1968, vol. 2, p. 283.
Stevens, W.J. and H. Basch, M., J. Krauss. Chem. Phys., 1984, vol. 81, p. 6026.
Laikov, D.N., Chem. Phys. Lett., 1997, vol. 281, p. 151.
Qu, L.J. and Jin, W.R., Anal. Chim. Acta, 1993, vol. 274, p. 65.
Farias, P.A., Martins, C.M.L., and Ohara, A.K., Anal. Chim. Acta, 1994, vol. 293, p. 29.
Mel’nikova, N.B., Ioffe, I.D., and Tsareva, L.A., Chemistry of Natural Compounds, 2002, vol. 38, p. 1.
Cotton, F.A. and Wilkinson, G., Advanced Inorganic Chemistry. A Comprehensive Text. 2nd edition, New York: Interscience, 1966.
Kuntic, V., Malesev, D., Radovic, Z., and Vukojevic, V., Monatshefte für Chemie, 2000, vol. 131, p. 769.
Afanas’ev, I.B., Ostrakhovitch, E.A., Mikhal’chik, E.A., Ibragimova, G.A., and Korkina, L.G., Biochem. Pharmac., 2001, vol. 61, p. 677.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © S.I. Kulakovskaya, L.A. Levchenko, A.P. Sadkov, N.G. Lobanova, A.F. Shestakov, 2009, published in Elektrokhimiya, 2009, Vol. 45, No. 10, pp. 1221–1231.
Rights and permissions
About this article
Cite this article
Kulakovskaya, S.I., Levchenko, L.A., Sadkov, A.P. et al. Electrochemical study of Au(III)-luteolin flavonoid system in tris-buffer. Russ J Electrochem 45, 1135–1144 (2009). https://doi.org/10.1134/S1023193509100048
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193509100048