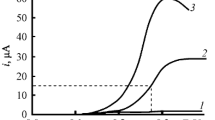
Abstract
Acetophenone and its intermediates formed upon the first electron transfer are studied by laser photoemission and traditional electrochemistry. It is shown that the intermediate reduction is affected by competition of the reactions of the formed radical-anions oxidation and their subsequent transformation to secondary products that are rapidly reduced at the electrode. From the comparison of the data obtained by the laser photoemission method and electrochemical measurements a conclusion was drawn that the product is a metastable complex (associate) of the radical-anion with water molecule; its formation rate constant is rather low (∼6 × 103 M−1 s−1). It was also concluded that bulk radical reactions dominate in aprotic media at moderate cathodic potentials; the acetophenone radical-anion is reduced at E ≤ −1.9 V (SCE). This conclusion agrees with the results of the acetophenone preparative electrolysis in DMFA, where marked yield of pinacon was observed at the potentials of limiting current of the 1st reduction wave, while the stage of 2nd electron transfer occurred at E ≤ −2.3 V (SCE).
Similar content being viewed by others
References
Organic Electrochemistry: An Introduction and a Guide, Baizer, M.M., Ed., New York: Marcel Dekker, 1973, Chapter 9.
Damaskin, B.B., Nekrasov, L.N., Petrii, O.A., Podlovchenko, B.I., Stenina, E.V., and Fedorovich, N.V., Elektrodnye protsessy v rastvorakh organicheskikh soedinenii (The Electrode Processes in Solutions of Organic Compounds), Moscow: Mosk. Gos. Univ., 1985.
Krivenko, A.G., Kotkin, A.S., Kurmaz, V.A., Elektrokhimiya, 2005, vol. 41, p. 157.
Krivenko, A.G. and Benderskii, V.A., Usp. Khim., 1990, vol. 59, p. 3.
Volke, J., Ryvolová-Kejharová, A. and Manoušek, O., J. Electroanal. Chem., 1971, vol. 32, p. 445.
Hayon, E., Ibata, T., Lichtin, N.N., and Simic, M., J. Phys. Chem., 1972, vol. 76, p. 2072.
Krivenko, A.G., Kotkin, A.S., Kurmaz, V.A., Elektrokhimiya, 2005, vol. 41, p. 142.
Mairanovskii, S.G., Beisembaeva, M.K., and Mambetkaziev, E.A., Elektrokhimiya, 1992, vol. 28, p. 1245.
Mousty, C. and Mousset, G., New J. Chem., 1992, vol. 16. p. 1063.
Ponomarev, E.A., Krivenko, A.G., Sviridova, L.N., and Stenina, E.V., Elektrokhimiya, 2001, vol. 37, p. 505.
Kodama, Y., Imoto, M., Ohta, N., Kitani, A., and Ito, S., J. Electroanal. Chem., 2001, vol. 507, p. 103.
Krivenko, A.G., Kotkin, A.S., Kurmaz, V.A., Rus. Khim. Zh., 2005, vol. 49, no. 5, p. 17.
Gultyai, V.P. and Rubinskaya, T.Ya., J. Electroanal. Chem., 1982, vol. 138, p. 185.
Nekrasov, L.N., Vykhodtseva, L.N., Korotaeva, L.M., and Gultyai, V.P., J. Electroanal. Chem., 1982, vol. 138, p. 177.
Rusakov, A.I., Mendkovich, A.S., Gultyai, V.P., and Orlov, V.Yu., Struktura i reaktsionnaya sposobnost’ organicheskikh anion-radikalov (Structure And Reactivity of Organic Anion-Radicals), Moscow: Mir, 2005.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.G. Krivenko, V.A. Kurmaz, A.S. Kotkin, G.V. Simbirtseva, V.P. Gultyai, 2008, published in Elektrokhimiya, 2008, Vol. 44, No. 12, pp. 1482–1485.
Rights and permissions
About this article
Cite this article
Krivenko, A.G., Kurmaz, V.A., Kotkin, A.S. et al. Generation of phenylmethylketyl radicals in aqueous solutions and study of their transformation. Russ J Electrochem 44, 1382–1385 (2008). https://doi.org/10.1134/S1023193508120112
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193508120112