Abstract
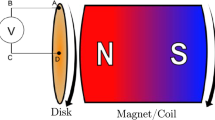
Based on the literature data and calculated results, the emf, which is generated in the rotating electrochemical cell with the electrodes located at different distances from the rotation axis, is estimated in the cases that the system is or is not placed in the magnetic field. These data enable one to determine the conditions (the angular cell rotational rate, the magnetic induction, etc.), under which the required emf can be obtained without an external power source. The application of magnetic field enables one increase the emf from several mV to several volts.
Similar content being viewed by others
References
Tolman, R.C., J. Am. Chem. Soc., 1911, vol. 33, p. 121.
Grinnell, S.W. and Koenig, F.O., J. Am. Chem. Soc., 1942, vol. 64, p. 682.
MacInnes, D.A. and Ray, B.R., J. Am. Chem. Soc., 1949, vol. 71, p. 2987.
Ray, B.R. and MacInnes, D.A., Rev. Sci. Inst., 1949, vol. 20, p. 52.
Ray, B.R., Beeson, D.M., and Crandall, H.F., J. Am. Chem. Soc., 1958, vol. 80, p. 1029.
MacInnes, D.A., The Principles of Electrochemistry, New York: Reinhold Publ. Corp, 1939, chapter 9.
Johnson, J.S., Kraus, K.A., and Young, T.F., J. Am. Chem. Soc., 1954, vol. 76, p. 1436.
Young, T.F., Kraus, K.A., and Johnson, J.S., J. Chem. Phys., 1954, vol. 22, p. 878.
Kegeles, G. and Narasinga, Rao M.S., J. Am. Chem. Soc., 1958, vol. 80, p. 5721.
Donnelly, T.H., J. Phys. Chem., 1966, vol. 70, p. 1862.
Godschalk, W., J. Phys. Chem., 1968, vol. 72, p. 498.
McBain, J.W., J. Am. Chem. Soc., 1936, vol. 58, p. 315.
Gokhshtein, A.Ya., Abstracts of Papers, VIII Mendeleevskii s’ezd po obshchei i prikladnoi khimii. Referaty dokladov i soobshcheniiπ 13. Sektsiya teoreticheskoi i prikladnoi elektrokhimii (VIII Mendeleev Conf. on General and Applied Chemistry. Section of Theoretical and Applied Electrochemistry), Moscow: Akad. Nauk SSSR, 1958, p. 65.
Thomson, E., US Patent 1701346, 1929.
Hoover, T.B., US Patent 3119759, 1964.
Cheng, H., Scott, K., and Ramshaw, C., J. Electrochem. Soc., 2002, vol. 149, p. D172.
Cheng, H., Scott, K., and Ramshaw, C., J. Appl. Electrochem., 2002, vol. 32, p. 831.
Kuiken, H.K. and Tijburg, R.P., J. Electrochem. Soc., 1983, vol. 130, p. 1722.
Tamminen, P., US Patent 4684585, 1987.
Atobe, M., Hitose, S., and Nonaka, T., Electrochem. Commun., 1999, vol. 1, p. 278.
Atobe, M., Sekido, M., Fuchigami, T., and Nonaka, T., Chem. Lett., 2003, p. 166.
Abakumov, G.A. and Fedoseev, V.B., Ross. Khim. Zh., 1998, vol. 42, p. 36.
Kolli, R., Zh. Russ. Khim. Fiz. O-va, Fiz. O-va, 1875, vol. 7, p. 333.
Colley, R., Ann. Phys. Chem, 1876, vol. 157, p. 624.
Des Coudres, T., Ann. Phys., 1893, vol. 49, p. 284.
Des Coudres, T., Ann. Phys., 1896, vol. 57, p. 232.
Tolman, R.C., Proc. Am. Acad. Arts Sci., 1910, vol. 46, p. 109.
Koenig, F.O. and Grinnell, S.W., J. Phys. Chem., 1940, vol. 44, p. 463.
Sandulov, D.B. and Doktorevich, V.A., Elektrokhimiya, 1984, vol. 20, p. 356.
Iwakura, C., Kitayama, M., Edamoto, T., and Tamura, H., Electrochim. Acta, 1985, vol. 30, p. 747.
Ghoroghchian, J. and Bockris, J.O’M., Int. J. Hydrogen Energy, 1985, vol. 10, p. 101.
Newman, J.S., Electrochemical Systems, New York: Prentice-Hall, 1973.
Pedersen, K.O., Z. Phys. Chem., 1934, vol. 170A, p. 41.
Drucker, C., Z. Phys. Chem., 1937, vol. 180A, p. 359.
Bograchev, D.A., Volgin, V.M., and Davydov, A.D., Elektrokhimiya, 2005, vol. 41, p. 1341 [Russ. J. Electrochem. (Engl. Transl.), vol. 41, p. 1197].
Feynman R.P., Leighton R.B., and Sands, M., The Feynman Lectures on Physics, London: Addison-Wesley, 1964, vol. 2.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D.A. Bograchev, A.D. Davydov, 2008, published in Elektrokhimiya, 2008, Vol. 44, No. 8, pp. 1029–1034.
Rights and permissions
About this article
Cite this article
Bograchev, D.A., Davydov, A.D. Generation of EMF in the rotating electrochemical cell. Russ J Electrochem 44, 956–961 (2008). https://doi.org/10.1134/S1023193508080120
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193508080120