Abstract
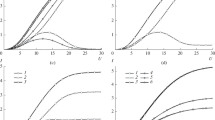
The process of mass transport during hydrogen ion reduction in aqueous hydrochloric acid solutions is examined both with and without excess supporting electrolyte. The study of this process is based on a numerical solution to a system of equations of material balance and the movement of particles in solution under the influence of forces for diffusion, migration, and convection. The homogeneous chemical reaction of water dissociation is also taken into account. The results of calculations show that a diffusion layer forms near the electrode during the passage of current in these solutions and that the effective thickness of this layer is the same at any instant for all particles participating in mass transport in solution in spite of differences in their diffusion coefficients. The value of the diffusion coefficient measured in these multicomponent solutions by the methods of chronopotentiometry and rotating disk electrode should differ little from that of hydrogen ions in spite of the fact that other particles with different diffusion coefficients participate in the mass transport.
Similar content being viewed by others
References
Levich, V.G., Fiziko-khimicheskaya gidrodinamika, 2nd ed., Moscow: Fizmatgiz, 1959 [Physicochemical Hydrodynamics (Engl. Transl.), Englewood Cliffs (NJ): Prentice-Hall, 1962].
Pleskov, Yu.V., and Filinovskii, V.Yu., Vrashchayushchiisya diskovyi elektrod (Rotating Disk Electrode), Moscow: Nauka, 1972.
Miyoshi, Y., and Lorenz, W.J., Ber. Bunsenges. Physik. Chem., 1970, vol. 74, p. 412.
Danauskas, A.V., Slizhis, R.P., and Matulis, Yu.Yu., Tr. Akad. Nauk Lit. SSR, Ser. B, 1973, vol. 4, no. 77, p. 3.
Riddiford, A.C., Advances in Electrochemistry and Electrochemical Engineering, New York: Interscience, 1966, vol. 4, ch. 2.
Newman, J.S., Electrochemical Systems, Englewood Cliffs (NJ): Prentice-Hall, 1973.
Pirogov, B.Ya., and Zelinsky, A.G., Electrochim. Acta, 2004, vol. 49, p. 3283.
Volmer, M., and Wick, H., Z. Phys. Chem., 1935, vol. 172A, p. 429.
Taylor, R., and Krishna, R., Multicomponent Mass Transfer, New York: Wiley, 1993, p. 95.
Ying, R.Y., Ng, P.K., Mao, Z., and White, R.E., J. Electrochem. Soc., 1988, vol. 135, p. 2964.
Vetter, K.J., Elektrochemische Kinetik, Berlin: Springer-Verlag, 1961.
Pirogov, B.Ya., and Zelinsky, A.G., J. Electroanal. Chem., 2006, vol. 593, p. 119.
Kharkats, Yu.I., Itogi nauki i tekhniki, Elektrokhimiya, 1991, vol. 38, p. 56.
Riddiford, A.C., and Gregory, D.P., J. Chem. Soc., 1956, p. 3756.
Damaskin, B.B., and Petrii, O.A., Vvedenie v elektrokhimicheskuyu kinetiku (Introduction to Electrochemical Kinetics), Moscow: Vysshaya shkola, 1975.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.G. Zelinsky, B.Ya. Pirogov, 2008, published in Elektrokhimiya, 2008, Vol. 44, No. 5, pp. 631–640.
The paper was prepared for special issue devoted to V.G. Levich 90th anniversary.
Rights and permissions
About this article
Cite this article
Zelinsky, A.G., Pirogov, B.Y. Effective thickness of the diffusion layer during hydrogen ion reduction in aqueous hydrochloric acid solutions. Russ J Electrochem 44, 585–593 (2008). https://doi.org/10.1134/S1023193508050133
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193508050133