Abstract
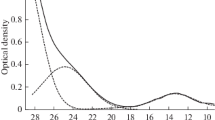
Products of anodic dissolution of uranium metal and the cathodic reduction of uranium(IV) ions in a molten eutectic mixture of lithium and potassium chlorides are studied at 450°C using x-ray absorption spectroscopy. It is shown that UCl 3−6 — ions form in the melt as a result of the metal’s anodic dissolution, and the metal-ligand distance in the complex ion is 0.284 nm. No complex ions of another configuration are found to form. Cathodic reduction of the UCl 2−6 — ions also yields the UCl 3−6 — ions, with longer U-Cl bonds.
Similar content being viewed by others
References
Kuznetsov, S.A., Hayashi, H., Minato, K., and Gaune-Escard, M. J. Electrochem. Soc., 2005, vol. 152, p. C203.
Meisel, A., Leonhardt, G., Szargan, R., X-Ray Spectra and Chemical Binding, Berlin: Springer, 1989.
Volkovich, V.A., May, I., Charnock, J.M., Rasplavy, 2004, no. 2, p. 76.
Volkovich, V.A., May, I., Charnock, J.M., and Griffiths, T.R., Rasplavy, 2006, no. 3, p. 48.
Okamoto, Y., Akabori, M., Itoh, A., and Ogawa, T., J. Nucl. Sci. Technol., 2002, suppl. 3, p. 638.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.A. Volkovich, I.B. Polovov, C.A. Sharrad, I. May, J.M. Charnock, 2007, published in Elektrokhimiya, 2007, Vol. 43, No. 8, pp. 1026–1030.
Rights and permissions
About this article
Cite this article
Volkovich, V.A., Polovov, I.B., Sharrad, C.A. et al. Uranium-involving electrode processes in chloride melts: An x-ray absorption spectroscopy study. Russ J Electrochem 43, 977–980 (2007). https://doi.org/10.1134/S1023193507080174
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1023193507080174