Abstract
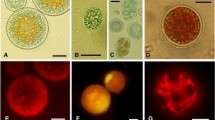
The relationships between changes in cell suspension absorbance, pigment composition, and resistance to photodamage were investigated in the microalga Haematococcus pluvialis Flotow em. Wille (Chlorophyta) IPPAS H-239 cultivated under high level of photosynthetically active radiation (PAR, 50 W/m2). When the green flagellated cells of H. pluvialis lacking astaxanthin (Ast) and possessing low (<0.5) carotenoid/chlorophyll ratio were irradiated by intense light (2500 W/m2 PAR), rapid and synchronous photobleaching of 70–80% of chlorophyll (Chl) and carotenoids (Car) was observed. By contrast, the rate of pigment photobleaching in cells with Car/Chl > 1, which retained high Chl content (> 0.6 fmol/cell) and accumulated significant amounts of Ast, was two times lower than in the green cells. Red aplanospores, with Car/Chl > 10, containing high amounts of Ast and low amounts of Chl (> 0.8 and < 0.1 fmol/cell, respectively) were resistant to photodestruction. The extent of cell resistance to photobleaching correlated closely with an increase in contribution of Car to light absorption by H. pluvialis cell suspensions. The build up of Ast during acclimation to high light was accompanied by a gradual increase in the optical density ratio OD480/OD678, whereas synchronous (OD480/OD678 ≈ const; r 2 > 0.99) and profound (>20%) bleaching of Car and Chl absorption bands was characteristic of photodamage. The spectral features of photoacclimation and photodamage revealed in this work can be used for nondestructive diagnostics of photodamage in H. pluvialis cultures and for on-line assessment of cell resistance to photooxidative death. The results are discussed with respect to the nondestructive monitoring of laboratory and production cultures of H. pluvialis and their protection from photooxidative death.
Similar content being viewed by others
Abbreviations
- Ast:
-
astaxanthin
- Car:
-
carotenoid(s)
- Chl:
-
chlorophyll(s)
- PAR:
-
photosynthetically active radiation
- PSA:
-
photosynthetic apparatus
- PUFA:
-
polyunsaturated fatty acids
- ROS:
-
reactive oxygen species
References
Bukhov, N.G., Dynamic Light Regulation of Photosynthesis (A Review), Russ. J. Plant Physiol., 2001, vol. 51, pp. 742–753.
Demmig-Adams, B. and Adams, W., Photoprotection in an Ecological Context: The Remarkable Complexity of Thermal Energy Dissipation, New Phytol., 2006, vol. 172, pp. 11–21.
Asada, K., Production and Scavenging of Reactive Oxygen Species in Chloroplasts and Their Functions, Plant Physiol., 2006, vol. 114, pp. 391–396.
Foyer, C. and Noctor, G., Oxygen Processing in Photosynthesis: Regulation and Signalling, New Phytol., 2000, vol. 146, pp. 359–388.
Hagen, C., Braune, W., and Björn, L., Functional Aspects of Secondary Carotenoids in Haematococcus lacustris (Volvocales): 3. Action as a Sunshade, J. Phycol., 1994, vol. 30, pp. 241–248.
Zhekisheva, M., Boussiba, S., Khozin-Goldberg, I., Zarka, A., and Cohen, Z., Accumulation of Oleic Acid in Haematococcus pluvialis (Chlorophyceae) under Nitrogen Starvation or High Light Is Correlated with That of Astaxanthin Esters, J. Phycol., 2002, vol. 38, pp. 325–331.
Wang, B., Zarka, A., Trebst, A., and Boussiba, S., Astaxanthin Accumulation in Haematococcus pluvialis (Chlorophyceae) as an Active Photoprotective Process under High Irradiance, J. Phycol., 2003, vol. 39, pp. 1116–1124.
Hu, Z., Li, Y., Sommerfeld, M., Chen, F., and Hu, Q., Enhanced Protection against Oxidative Stress in an Astaxanthin-Overproduction Haematococcus Mutant (Chlorophyceae), Eur. J. Phycol., 2008, vol. 43, pp. 365–376.
Solovchenko, A.E., Khozin-Goldberg, I., DidiCohen, S., Cohen, Z., and Merzlyak, M.N., Effects of Light and Nitrogen Starvation on the Content and Composition of Carotenoids of the Green Microalga Parietochloris incisa, Russ. J. Plant Physiol., 2008, vol. 55, pp. 455–462.
Bar, E., Rise, M., Vishkautsan, M., and Arad, S., Pigment and Structural Changes in Chlorella zofingiensis upon Light and Nitrogen Stress, J. Plant Physiol., 1995, vol. 146, pp. 527–534.
Hanagata, N. and Dubinsky, Z., Secondary Carotenoid Accumulation in Scenedesmus komarekii (Chlorophyceae, Chlorophyta), J. Phycol., 1999, vol. 35, pp. 960–966.
Ben-Amotz, A., Shaish, A., and Avron, M., Mode of Action of the Massively Accumulated β-Carotene of Dunaliella bardawil in Protecting the Alga against Damage by Excess Irradiation, Plant Physiol., 1989, vol. 86, pp. 1286–1291.
Rabbani, S., Beyer, P., Lintig, J., Hugueney, P., and Kleinig, H., Induced β-Carotene Synthesis Driven by Triacylglycerol Deposition in the Unicellular Alga Dunaliella bardawil, Plant Physiol., 1998, vol. 116, pp. 1239–1248.
Solovchenko, A.E. and Merzlyak, M.N., Screening of Visible and UV Radiation as a Photoprotective Mechanism in Plants, Russ. J. Plant Physiol., 2008, vol. 55, pp. 719–737.
Torzillo, G., Goksan, T., Faraloni, C., Kopecky, J., and Masojdek, J., Interplay between Photochemical Activities and Pigment Composition in an Outdoor Culture of Haematococcus pluvialis during the Shift from the Green to Red Stage, J. Appl. Phycol., 2003, vol. 15, pp. 127–136.
Fabregas, J., Dominguez, A., Maseda, A., and Otero, A., Interactions between Irradiance and Nutrient Availability during Astaxanthin Accumulation and Degradation in Haematococcus pluvialis, Appl. Microbiol. Biotechnol., 2003, vol. 61, pp. 545–551.
Sussela, M. and Toppo, K., Haematococcus pluvialis—a Green Alga, Richest Natural Source of Astaxanthin, Curr. Sci., 2006, vol. 90, pp. 1602–1603.
Vidhyavathi, R., Venkatachalam, L., Sarada, R., and Ravishankar, G., Regulation of Carotenoid Biosynthetic Genes Expression and Carotenoid Accumulation in the Green Alga Haematococcus pluvialis under Nutrient Stress Conditions, J. Exp. Bot., 2008, vol. 59, pp. 1409–1418.
Boussiba, S., Carotenogenesis in the Green Alga Haematococcus pluvialis: Cellular Physiology and Stress Response, Physiol Plant., 2000, vol. 108, pp. 111–117.
Pratt, R., Studies on Chlorella vulgaris. XI. Relation between Surface Tension and Accumulation of Chlorellin, Am. J. Bot., 1948, vol. 35, pp. 634–637.
Wellburn, A., The Spectral Determination of Chlorophyll a and Chlorophyll b, as Well as Total Carotenoids, Using Various Solvents with Spectrophotometers of Different Resolution, J. Plant Physiol., 1994, vol. 144, pp. 307–313.
Merzlyak, M.N., Chivkunova, O.B., Maslova, I.P., Naqvi, K.R., Solovchenk, o A.E., and Klyachko-Gurvich, G.L., Light Absorption and Scattering by Cell Suspensions of Some Cyanobacteria and Microalgae, Russ. J. Plant Physiol., 2008, vol. 55, pp. 420–425.
Czygan, F., Blood-Rain and Blood-Snow: Nitrogen-Deficient Cells of Haematococcus pluvialis and Chlamydomonas nivalis, Arch. Mikrobiol., 1970, vol. 74, pp. 69–76.
Kobayashi, M. and Sakamoto, Y., Singlet Oxygen Quenching Ability of Astaxanthin Esters from the Green Alga Haematococcus pluvialis, Biotechnol. Lett., 1999, vol. 21, pp. 265–269.
Kobayashi, M., In Vivo Antioxidant Role of Astaxanthin under Oxidative Stress in the Green Alga Haematococcus pluvialis, Appl. Microbiol. Biotechnol., 2000, vol. 54, pp. 550–555.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.E. Solovchenko, O.B. Chivkunova, I.P. Maslova, 2011, published in Fiziologiya Rastenii, 2011, Vol. 58, No.1, pp. 12–20.
Rights and permissions
About this article
Cite this article
Solovchenko, A.E., Chivkunova, O.B. & Maslova, I.P. Pigment composition, optical properties, and resistance to photodamage of the microalga Haematococcus pluvialis cultivated under high light. Russ J Plant Physiol 58, 9–17 (2011). https://doi.org/10.1134/S1021443710061056
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443710061056