Abstract
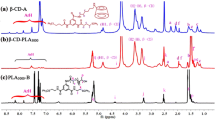
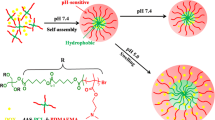
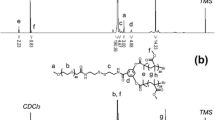
The surface cRGD modified polymeric (PMMA-b-PPEGMA-RGD) micelles were constructed by a combination of two-step atom transfer radical polymerization (ATRP), azide reaction, click reaction, deprotection, self-assembly of amphiphilic polymers, and addition reaction. The self-assembled DOX-loaded micelles had spherical morphology. The average size of DOX-loaded micelles measured by scanning electron microscopy (SEM) and dynamic light scattering (DLS) was less than 200 nm. According to the enhanced permeability and retention (EPR) effect of solid tumors, the micelles should have the passive targeting of tumor tissues. The introduction of cRGD on the micelles surface can not only achieve the active targeting of drug carriers to tumor tissues, but also form the network hydrogen bond between the PEG unit at the hydrophilic end of the micelles and the guanidine group in RGD, to improve the uniformity of the particle size distribution and the stability of drug loading, and also achieve the slow release of drugs. The cytotoxicity of DOX-loaded micelles on SGC7901 cells in vitro indicated that PMMA-b-PPEGMA-RGD micelles could provide the same or more remarkable antitumor activity (at low dose) as DOX. The results showed that PMMA-b-PPEGMA-RGD micelles might have great potential applications in the delivery of hydrophobic anticancer drugs.





Similar content being viewed by others
REFERENCES
K. S. Soppimatha, T. M. Aminabhavi, A. R. Kulkarni, and W. E. Rudzinski, J. Controlled Release 70, 1 (2001).
D. Peer, J. M. Karp, S. Hong, O. C. Farokhzad, R. Margalit, and R. Langer, Nat. Nanotechnol. 2, 751 (2007).
T. M. Allen and P. R. Cullis, Science 303, 1818 (2004).
Z. X. You, Q. Z. Yang, Z. G. Yue, and G. H. Ma, Chin. J. Tissue Eng. Res. 16, 4701 (2012).
P. C. Brooks, R. A. Clark, and D. A. Cheresh, Science 264, 569 (1994).
A. Adulnirath, S. W. Chung, J. Park, S. R. Hwang, J. Y. Kim, V. C. Yang, S. Y. Kim, H. T. Moon, and Y. Byun, J. Controlled Release 164, 8 (2012).
Q. Cao, Z. B. Li, C. Kai, Z. Wu, L. He, N. Neamati, and X. Chen, Eur. J. Nucl. Med. Mol. Imaging 35, 1489 (2008).
X. B. Xiong, Y. Huang, W. L. Lu, X. Zhang, H. Zhang, T. Nagai, and Q. Zhang, J. Pharm. Sci. 94, 1782 (2005).
C. L. Waite and C. M. Roth, Bioconjugate Chem. 20, 1908 (2009).
T. D. Le, O. Nakagawa, M. Fisher, R. L. Juliano, and H. Yoo, J. Nanosci. Nanotechnol. 17, 2353 (2017).
C. Cui, Y. Wang, K. Yang, Y. Wang, J. Yang, J. Xi, M. Zhao, J. Wu, and S. Peng, J. Biomed. Nanotechnol. 11, 70 (2015).
J. Jiang, W. Wang, D. C. Sane, and B. Wang, Bioorg. Chem. 29, 357 (2001).
X. Jiang, X. Fan, W. Xu, C. Zhao, H. Wu, R. Zhang, and G. Wu, J. Controlled Release 316, 196 (2019).
H. F. Wang, H. Z. Jia, S. X. Cheng, J. Feng, X. Z. Zhang, and R. X. Zhuo, Pharm. Res. 29, 1582 (2012).
Y. I. Jeong, H. S. Na, K. O. Cho, H. C. Lee, J. W. Nah, and C. S. Cho, Int. J. Pharm. 365, 150 (2009).
S. W. Hong, K. H. Kim, J. Huh, C. H. Ahn, and W. H. Jo, Macromol. Res. 13, 397 (2005).
M. Kawahata, K. Yamaguchi, and T. Ishikawa, Cryst. Growth Des. 5, 373 (2005).
J. Jo, A. Olasz, C. H. Chen, and D. Lee, J. Am. Chem. Soc. 135, 3620 (2013).
R. Frański, G. Schroeder, B. Gierczyk, P. Niedziałkowski, and T. Ossowski, Int. J. Mass Spectrom. 266, 180 (2007).
J. Geng, G. Mantovani, L. Tao, J. Nicolas, G. Chen, R. Wallis, D. A. Mitchell, B. R. G. Johnson, S. D. Evans, and D. M. Haddleton, J. Am. Chem. Soc. 129, 15156 (2007).
B. Y. Zhang, W. D. He, W. T. Li, L. Y. Li, K. R. Zhang, and H. Zhang, Polymer 51, 3039 (2010).
A. Späth and B. König, Tetrahedron 66, 6019 (2010).
Funding
This work was financially supported by the Key Research and Development Program of Shaanxi (no. 2021ZDLSF03-05), Scientific and technological innovation team of Xi’an Medical College (2021DT07), the Youth Support Scheme Project of Shaanxi University Association for science and technology (no. 20170410), Xi’an Science and technology plan project (2020KJRC0135), Xi’an Weiyang District Science and technology plan project (201930) and Science Research Program of Xi’an Medical University (nos. 2017GJFY07, 2018PT65, 05041905, 2016YXXK03, 201906).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Supplementary Information
Rights and permissions
About this article
Cite this article
Yu, L., Yao, L., Yang, K. et al. Study on the Preparation and Properties of Highly Stable Micelles Sealed by Hydrogen Bonds. Polym. Sci. Ser. A 63, 769–778 (2021). https://doi.org/10.1134/S0965545X21350169
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965545X21350169