Abstract
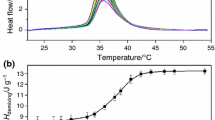
It is shown that aqueous solutions of polyethylaminophosphazene hydrochloride undergo phase separation during heating. This phenomenon is studied in detail at pH 3.5 (0.1 M citrate buffer) in relation to the composition of the system with the use of nephelometry and high-sensitivity DSC. The cloud points and the enthalpy of phase separation of the system are determined, and its phase diagram is constructed. The system features a lower critical solution temperature: w 2,cr = 7.3 × 10−4 and T cr = 34.3°C. The enthalpy of phase separation is 12.8 ± 0.6 J/g of the polymer, regardless of the system composition. A new approach to the analysis of DSC data on the phase separation of aqueous solutions of polymers during heating is advanced on the basis of calorimetric parameters coupled with the data on the composition of coexisting phases. Through this approach, the main contribution to the heat effect of phase separation of the system under study is related to a change in the energy state of a polyethylaminophosphazene hydrochloride molecule as a result of its dehydration.
Similar content being viewed by others
References
S. P. Papkov, Phase Equilibrium in Polymer-Solvent Systems (Khimiya, Moscow, 1981) [in Russian].
S. P. Papkov, Physicochemical Principles of Polymer Solution Processing (Khimiya, Moscow, 1971) [in Russian].
R. X. Liu, M. Fraylich, and B. R. Saunders, Colloid Polym. Sci. 287, 627 (2009).
B. Jeong and A. Gutowska, Trends Biotechnol. 20, 305 (2002).
E. S. Gil and S. A. Hudson, Prog. Polym. Sci. 29, 1173 (2004).
M. Hruby, J. Kucka, H. Mackova, et al., Chem. Listy 102, 21 (2008).
L. Klouda and A. G. Mikos, Eur. J. Pharm. Biopharm. 68, 34 (2008).
S. M. Moghimi and A. C. Hunter, Trends Biotechnol. 18, 412 (2000).
B. Jeong, Y. H. Bae, D. S. Lee, and S. W. Kim, Nature (London) 388, 860 (1997).
M. V. Chaubal, A. S. Gupta, S. T. Lopina, and D. F. Bruley, Crit. Rev. Ther. Drug Carrier Syst. 20, 295 (2003).
A. C. Couffin-Hoarau and J. C. Leroux, Biomacromolecules 5, 2082 (2004).
G. Ilia, Polym. Adv. Technol. 20, 707 (2009).
M. K. Joo, M. H. Park, B. G. Choi, and B. Jeong, J. Mater. Chem. 19, 5891 (2009).
G. D. Kang, S. H. Cheon, G. Khang, and S. C. Song, Eur. J. Pharm. Biopharm. 63, 340 (2006).
S. Lakshmi, D. S. Katti, and C. T. Laurencin, Adv. Drug Delivery Rev. 55, 467 (2003).
J. Luten, J. H. Van Steenis, R. Van Someren, et al., J. Controlled Release 89, 483 (2003).
S. G. Kumbar, S. Bhattacharyya, S. P. Nukavarapu, et al., J. Inorg. Organomet. Polym. Mater. 16, 365 (2006).
V. Ya. Grinberg, T. V. Burova, N. V. Grinberg, et al., Polymer Science, Ser. A 51, 390 (2009) [Vysokomol. Soedin., Ser. A 51, 591 (2009)].
D. R. Tur, D. V. Pergushov, I. A. Babin, et al., Polymer Science, Ser. B 51, 360 (2009) [Vysokomol. Soedin., Ser. B 51, 1704 (2009)].
H. R. Allcock, S. R. Pucher, M. L. Turner, and R. J. Fitzpatrick, Macromolecules 25, 5573 (1992).
H. R. Allcock and G. K. Dudley, Macromolecules 29, 1313 (1996).
S. B. Lee, S. C. Song, J. I. Jin, and Y. S. Sohn, Macromolecules 32, 7820 (1999).
S. B. Lee, S. C. Song, J. I. Jin, and Y. S. Sohn, Colloid Polym. Sci. 278, 1097 (2000).
S. B. Lee, S. C. Song, J. I. Jin, and Y. S. Sohn, Polym. Bull. (Berlin) 45, 389 (2000).
S. B. Lee, Y. S. Sohn, and S. C. Song, Bull. Korean Chem. Soc. 24, 901 (2003).
S. Ahn, S. W. Ahn, and S. C. Song, Colloids Surf., A 330, 184 (2008).
S. Ahn, E. C. Monge, and S. C. Song, Langmuir 25, 2407 (2009).
S. C. Song, S. B. Lee, J. I. Jin, and Y. S. Sohn, Macromolecules 32, 2188 (1999).
J. Y. Yoon, K. H. Park, and S. C. Song, J. Biomater. Sci., Polym. Ed. 18, 1181 (2007).
S. M. Lee, C. J. Chun, J. Y. Heo, and S. C. Song, J. Appl. Polym. Sci. 113, 3831 (2009).
T. Potta, C. Chun, and S. C. Song, Biomaterials 30, 6178 (2009).
P. L. Privalov and S. A. Potekhin, Methods Enzymol. 131, 4 (1986).
H. G. Schild, Prog. Polym. Sci. 17, 163 (1992).
A. S. Dubovik, E. E. Makhaeva, V. Ya. Grinberg, and A. R. Khokhlov, Macromol. Chem. Phys. 206, 915 (2005).
Y. Okada and F. Tanaka, Macromolecules 38, 4465 (2005).
R. Koningsveld and A. J. Staverman, J. Polym. Sci., Part A-2 6, 325 (1968).
R. Koningsveld and A. J. Staverman, J. Polym. Sci., Part A-2 6, 349 (1968).
A. S. Dubovik, N. V. Grinberg, and V. Ya. Grinberg, Polymer Science, Ser. A 52, 565 (2010) [Vysokomol. Soedin., Ser. A 52, 899 (2010)].
J. Arnauts, R. Decooman, P. Vandeweerdt, et al., Thermochim. Acta 238, 1 (1994).
C. Tanford, Physical Chemistry of Macromolecules (Wiley, New York, 1961), p. 204.
V. Ya. Anosov, M. I. Ozerova, and Yu. Ya. Fialkov, Fundamentals of Physicochemical Analysis (Nauka, Moscow, 1978) [in Russian].
C. Tanford, Physical Chemistry of Macromolecules (Wiley, New York, 1961), p. 245.
Z. Ahmed, E. A. Gooding, K. V. Pimenov, et al., J. Phys. Chem. B 113, 4248 (2009).
I. Prigogine and R. Defay, Chemical Thermodynamics (Longmans, London, 1954; Nauka, Novosibirsk, 1966).
H. Durchschlag, in Thermodynamic Data for Biochemistry and Biotechnology, Ed. by H. J. Hinz (Springer, Berlin, 1986), p. 45.
J. Y. Seong, Y. J. Jun, B. M. Kim, et al., Int. J. Pharm. 314, 90 (2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.Ya. Grinberg, N.V. Grinberg, T.V. Burova, A.S. Dubovik, D.R. Tur, V.S. Papkov, 2010, published in Vysokomolekulyarnye Soedineniya, Ser. A, 2010, Vol. 52, No. 11, pp. 2025–2033.
This work was supported by the Russian Foundation for Basic Research (project no. 07-03-00476-a) and by the Division of Chemistry and Materials Sciences, Russian Academy of Sciences, under the program “Creation and Study of Macromolecules and Macromolecular Structures of New Generations.”
Rights and permissions
About this article
Cite this article
Grinberg, V.Y., Grinberg, N.V., Burova, T.V. et al. Phase separation in aqueous solutions of polyethylaminophosphazene hydrochloride during heating. Polym. Sci. Ser. A 52, 1220–1227 (2010). https://doi.org/10.1134/S0965545X10110167
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965545X10110167