Abstract
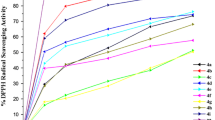
A number of new derivatives of N-benzylarylamines and diphenyl α-aminomethanephosphonates have been synthesized, and their structure has been established by IR and 1H NMR spectroscopy. It has been shown that these compounds can be used as effective additives enhancing the thermooxidative stability of ester-based oils.
Similar content being viewed by others
References
A. M. Kuliev, Chemistry and Technology of Oil and Fuel Additives (Khimiya, Leningrad, 1985) [in Russian].
M. P. Fieser, Fieser’s Reagents for Organic Synthesis, (Wiley, New York, 1979), vol. 7.
G. Hilgetag and A. Martini, Weygand-Hilgetag organisch-chemische Experimentierkunst (Johann Ambrosius Barth: Leipzig, 1964; Khimiya, Moscow, 1968), 3rd ed. p. 944.
Yu. A. Bruk, F. Yu. Rachinskii, L. V. Zolotova, et al., Zh. Obshch. Khim. 42 (1972).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.K. Kyazim-zade, A.A. Gadirov, 2011, published in Neftekhimiya, 2011, Vol. 51, No. 6, pp. 473–476.
Rights and permissions
About this article
Cite this article
Kyazim-zade, A.K., Gadirov, A.A. Synthesis of N-benzylarylamine derivatives and diphenyl esters of α-aminomethanephosphonic acids and examination of their antioxidant properties. Pet. Chem. 51, 465–468 (2011). https://doi.org/10.1134/S0965544111060119
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965544111060119