Abstract
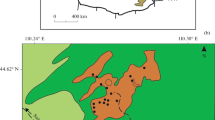
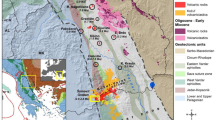
The paper presents mineralogical and geochemical data on clinkers and paralavas and on conditions under which they were formed at the Nyalga combustion metamorphic complex, which was recently discovered in Central Mongolia. Mineral and phase assemblages of the CM rocks do not have analogues in the world. The clinkers contain pyrogenically modified mudstone relics, acid silicate glass, partly molten quartz and feldspar grains, and newly formed indialite microlites (phenocrysts) with a ferroindialite marginal zone. In the paralava melts, spinel microlites with broadly varying Fe concentrations and anorthite–bytownite were the first to crystallize, and were followed by phenocrysts of Al-clinopyroxene ± melilite and Mg–Fe olivine. The next minerals to crystallize were Ca-fayalite, kirschsteinite, pyrrhotite, minerals of the rhönite–kuratite series, K–Ba feldspars (celsian, hyalophane, and Ba-orthoclase, Fe3+-hercynite ± (native iron, wüstite, Al-magnetite, and fresnoite), nepheline ± (kalsilite), and later calcite, siderite, barite, celestine, and gypsum. The paralavas contain rare minerals of the rhönite–kuratite series, a new end-member of the rhönite subgroup Ca4Fe 2+8 Fe 3+4 O4 [Si8Al4O36], a tobermorite-like mineral Ca5Si5(Al,Fe)(OH)O16 · 5H2O, and high- Ba F-rich mica (K,Ba)(Mg,Fe)3(Al,Si)4O10F2. The paralavas host quenched relics of microemulsions of immiscible residual silicate melts with broadly varying Si, Al, Fe, Ca, K, Ba, and Sr concentrations, sulfide and calcitic melts, and water-rich silicate–iron ± (Mn) fluid media. The clinkers were formed less than 2 Ma ago in various parts of the Choir–Nyalga basin by melting Early Cretaceous mudstones with bulk composition varies from dacitic to andesitic. The pyrogenic transformations of the mudstones were nearly isochemical, except only for volatile components. The CM melt rocks of basaltic andesitic composition were formed via melting carbonate–silicate sediments at temperatures above 1450°C. The Ca- and Fe-enriched and silicaundersaturated paralavas crystallized near the surface at temperatures higher than 900–1100°C and oxygen fugacity \(f_{O_2 }\) between the IW and QFM buffers. In local melting domains of the carbonate–silicate sedimentary rocks and in isolations of the residual melts among the paralava matrix the fluid pressure was higher than the atmospheric one. The bulk composition, mineral and phase assemblages of CM rocks of the Nyalga complex are very diverse (dacitic, andesitic, basaltic andesitic, basaltic, and silica-undersaturated mafic) because the melts crystallized under unequilibrated conditions and were derived by the complete or partial melting of clayey and carbonate–silicate sediments during natural coal fires.
Similar content being viewed by others
References
Balassone, G., Franco, E., Mattia, C.F., and Pulity, R., Indialite in xenolitic rocks from Somma–Vesuvius volcano (southern Italy): crystal chemistry and petrogenetic features, Am. Mineral., 2004, vol. 89, pp. 1–6.
Bentor, Y.K., Gross, S., and Heller, L., High-temperature minerals in non-metamorphosed sediments in Israel, Nature, 1963, vol. 199, pp. 478–479.
Bentor, Y.K., Kastner, M., Perlman, I., and Yellin, L., Combustion metamorphism of bituminous sediments and the formation of melts of granitic and sedimentary composition, Geochim. Cosmochim. Acta, 1981, vol. 45, pp. 2229–2255.
Bentor, Y.K., Combustion metamorphic glasses, J. Non-Cryst. Solids, 1984, vol. 67, pp. 433–448.
Boivin, P., Données expérimentales préliminaires sur la stabilité de la rhönite à 1 atmosphère. Application aux gisements naturels, Bull. Mineral., 1980, vol. 103, pp. 491–502.
Bratash, V.I., Report of party No. 26 on Prospecting Works in 1951 on a Scale 1: 200000 in the Eastern Part of the Nyalga Basin of the Central Aimak of Mongolia, Ulan-Bator, 1952, Ulan-Bator: Geological Fund,, Cadastre no. 1842.
Bratash, V.I. and Novikova, L.A., Report on 1: 200000 geological survey in the Nyalga Basin, Ulan-Bator, 1952–1953, Ulan-Bator: Geological Fund, Cadastre No. 1309.
Burg, A., Kolodny, Ye., and Lyakhovsky, V., Hatrurim- 2000: the “mottled” “zone” revisited, forty years later, Isr. J. Earth Sci., 1999, vol. 48, pp. 209–223.
Chukanov, N.V., Aksenov, S.M., Pekov, I.V., et al., Ferroindialite (Fe2+,Mg)2Al4Si5O18—a new beryl group mineral from the Eifel volcanic area, Germany, Zap. Ross. Mineral. O-va, 2014, vol. 143, no. 1, pp. 46–56.
Coal and Peat Fires: a Global Perspective. Volume 3. Case Studies and Coal Fires, Stracher, G.B., Prakash, A., and Sokol, E.V., Eds., Amsterdam: Elsevier, 2015.
Cosca, M. and Peacor, D., Chemistry and structure of esseneite, (CaFe3+AlSiO6), a new pyroxene produced by pyrometamorphism, Am. Mineral., 1987, vol. 72, pp. 148–156.
Cosca, M.A., Essene, E.J., Geissman, J.G., et al., Pyrometamorphic rocks associated with naturally burned coal beds, Powder River basin, Wyoming, Am. Mineral., 1989, vol. 74, pp. 85–100.
Davidson, P.M. and Mukhopadhyay, D.K., Ca–Fe–Mg olivines: phase relations and a solution model, Contrib. Mineral. Petrol., 1984, vol. 86, pp. 256–263.
Deer, W.A., Howie, R.A., and Zussman, J., An Introduction to the Rock-Forming Minerals, Essex–New York: Longman Scientific and Technical–Wiley, 1992.
Durand, C., Baumgartner, L.P., and Marquer, D., Low melting temperature for calcite at 1000 bars on the join CaCO3-H2O—some geological implications, Terra Nova, 2015, vol. 27, pp. 364–369.
Erdenetsogt, B., Lee, I., Bat-Erdene, D., and Jargal, L., Mongolian coal-bearing basins: geological settings, coal characteristics, distribution, and resources, Int. J. Coal Geol., 2009, vol. 80, pp. 87–104.
Foit, F.F., Hooper, R.L., and Rosenberg, P.E., An unusual pyroxene, melilite, and iron oxide mineral assemblage in a coal-fire buchite from Buffalo, Wyoming, Am. Mineral., 1987, vol. 72, pp. 137–147.
Genshaft, Yu.S. and Saltykovskii, A.Ya. Cenozoic volcanism of Mongolia, Ross. Zh. Nauk Zemle, 2000, vol. 2, no. 2, pp. 153–183.
Genshaft, Yu.S., Klimenko, G.V., Saltykovskii, A.Ya., Ageeva, L.I., New data on composition and age of Cenozoic volcanic rocks of Mongolia, Dokl. Akad. Nauk SSSR, 1990, vol. 311, no. 2, pp. 420–424.
Gittins, J. and Tuttle, O.F., The system CaF2-Ca(OH)2- CaCO3, Am. J. Sci., 1964, vol. 262, pp. 66–75.
Grapes, R., Pyrometamorphism, Berlin: Springer, 2011.
Grapes, R. and Keller, J., Fe2+-dominant rhönite in undersaturated alkaline basaltic rocks, Kaiserstuhl volcanic complex, Upper Rhine graben, SWGermany, Eur. J. Mineral, 2010, vol. 22, pp. 285–292.
Grapes, R., Zhang, K., and Peng, Z., Paralava and clinker products of coal combustion, Yellow River, Shanxi Province, China, Lithos, 2009, vol. 113, pp. 831–843.
Grapes, R., Korzhova, S., Sokol, E., and Seryotkin, Y., Paragenesis of unusual Fe-cordierite (sekaninaite)-bearing paralava and clinker from the Kuznetsk coal basin, Siberia, Russia, Contrib. Mineral. Petrol., 2011, vol. 162, pp. 253–273.
Grapes, R., Sokol, E., Kokh, S., et al., Petrogenesis of Narich paralava formed by methane flares associated with mud volcanism, Altyn-Emel national park, kazakhstan, Contrib. Mineral. Petrol., 2013, vol. 165, pp. 781–803.
Grew, E.S., Hålenius, U., Pasero, M., and Barbier, J., Recommended nomenclature for the sapphirine and surinamite groups (sapphirine supergroup), Mineral. Mag., 2008, vol. 72, pp. 39–876.
Haefeker, U., Kaindl, R., and Tropper, P., Semi-quantitative determination of the Fe/Mg ratio in synthetic cordierite using Raman spectroscopy, Am. Mineral., 2012, vol. 97, pp. 1662–1669.
Haggerty, S.E., Oxide mineralogy of the upper mantle, in Oxide Minerals: Petrologic and Magnetic Significance, Lindsley, D.H., Eds., Mineral. Soc. Amer. Rev. Mineral., 1991, vol. 25, pp. 355–416.
He, Y.T. and Traina, S.J., Transformation of magnetite to goethite under alkaline Ph conditions, Clay Mineral., 2007, vol. 42, pp. 13–19.
Heffern, E.L., Reiners, P.W., Naeser, C.W., and Coates, D.A., Geochronology of clinker and implications for evolution of the Powder River basin landscape, Wyoming and Montana, Geol. Soc. Amer. Rev. Eng. Geol., 2007, pp. 155–175.
Hess, J.C. and Lippolt, H.J., Compilation of K-Ar measurements on HD-B1 standard biotite–1994 status report, in Phanerozoic Time Scalel, Odin, G.S., Eds., Bul. Liasis. Inform. IUGS Subcom. Geochronol., 1994, vol. 12, pp. 19–23.
Hwang, S-L., Shen, P., Chu, H-T., et al., Kuratite Ca4(Fe2+ 10Ti2)O4[Si8Al4O36], the Fe2+-analogue of rhönite, a new mineral from D’Orbigny angrite meteorite, Mineral. Mag., 2016, vol. 80, pp. 1067–1076.
Kalugin, I.A., Tret’yakov, G.A., and Bobrov, V.A., Iron ore basalts in the burnt rocks of East Kazakhstan, Tr. Inst. Geol. Geofiz. Sib. Otd. Akad. Nauk SSSR, Novosibirsk, 1991.
Keller, J., Zaitsev, A.N., and Wiedenmann, D., Primary magmas at Oldoinyo Lengai: the role of olivine melilitites, Lithos, 2006, vol. 91, pp. 150–172.
Kunzmann, T., The aenigmatite–rhönite mineral group, Eur. J. Mineral., 1999, vol. 11, pp. 743–756.
Lavrent’ev, Yu.G., Karmanov, N.S., and Usova, L.V. Electron probe microanalysis of minerals: microprobe or scanning electron microscope? Russ. Geol. Geophys., 2015, vol. 56, no. 8, pp. 1154–1161.
Lee, J.-Y., Marti, K., Severinghaus, J.P., et al., A redetermination of the isotopic abundances of atmospheric Ar, Geochim. Cosmochim. Acta, 2006, vol. 70, pp. 4507–4512.
McDonough, W.E. and Sun, S., The composition of the Earth, Chem. Geol., 1995, vol. 120, pp. 223–253.
Melluso, L., Conticelli, S., and Gennaro, R., Kirschsteinite in the Capo di Bove melilite leucitite lava (cecilite), Alban Hills, Italy, Mineral. Mag., 2010, vol. 74, pp. 887–902.
Mukhopadhyay, D.K. and Lindsley, D.H., Phase relations in the join kirschsteinite (CaFeSiO4)–fayalite (Fe2SiO4), Am. Mineral., 1983, vol. 68, pp. 1089–1094.
Nigmatulina, E.N. and Nigmatulina, E.A., Pyrogenic iron ores of ancient coal fires of the Kuznetsk Basin, Zap. Ross. Mineral. O-va, 2009, no. 1, pp. 52–68.
Novikov, I.S., Sokol, E.V., Travin, A.V., and Novikova, S.A., Signature of Cenozoic orogenic movements in combustion metamorphic rocks: mineralogy and geochronology (example of the Salair–Kuznetsk Basin transition), Russ. Geol. Geophys., 2008, vol. 49, no. 6, pp. 378–396.
Novikova, S., Sokol, E., and Khvorov, P., Multiple combustion metamorphic events in the Goose Lake coal basin, Transbaikalia, Russia: first dating results, Quat. Geochronol., 2016, vol. 36, pp. 38–54.
Peretyazhko, I.S., CRYSTAL–Applied software for mineralogists, petrologists, and geochemists, Zap. Ross. Mineral. O-va, 1996, no. 3, pp. 141–148.
Peretyazhko, I.S. and Savina, E.A., Silicate–iron liquid immiscibility in rhyolitic magma, Dokl. Earth Sci., 2014, vol. 457, pp. 1028–1033.
Peretyazhko I.S., Savina E.A., Karmanov N.S., and Pavlova, L.A., Silicate–iron fluid media in rhyolitic magma: data on rhyolites from the Nilginskaya Depression, Central Mongolia, Petrology, 2014, vol. 22, no. 3, pp. 255–292.
Peretyazhko, I.S., Savina, E.A., and Khromova, E.A., Minerals of the rhönite–kuratite series in paralavas from a new combustion metamorphic complex of Choir–Nyalga basin (Central Mongolia): chemistry, mineral assemblages, and formation conditions, Mineral. Mag., 2017, vol. 81, pp. 949–974.
Platz, T., Foley, S.F., and André, L., Low-pressure fractionation of the Nyiragongo volcanic rocks, Virunga Province, D.R. Congo, J. Volcanol. Geotherm. Res., 2004, vol. 136, pp. 269–295.
Pokrovskii, P.V., Ammonium chloride from the Khamarin–Khural–Khid brown coal field in Mongolia, Zap. Ross. Mineral. O-va, 1949, no. 3, pp. 38–45.
Richardson, I.G., Tobermorite/jennite- and tobermorite/calcium hydroxide-based models for the structure of C–S–H: applicability to hardened pastes of tricalcium silicate, ß-dicalcium silicate, Portland cement, and blends of Portland cement with blast-furnace slag, metakaolin, or silica fume, Cement Concrete Res., 2004, vol. 34, pp. 1733–1777.
Schreyer, W., Maresch, W.V., Daniels, P., and Wolfsdorff, P., Potassic cordierite: characteristic minerals for high-temperature, very low-pressure environments, Contrib. Mineral. Petrol., 1990, vol. 105, pp. 162–172.
Sharpenok, L.N., Kukharenko, E.A., and Kostin, A.E., New provisions for volcanogenic rocks in the petrographic code, J. Volcanol. Seismol., 2009, vol. 3, no. 4, pp. 279–293.
Sharygin, V.V., Sokol E.V., and Belakovskii, D.I., Fayalite–sekaninaite paralava from the Ravat coal fire (Central Tajikistan), Russ. Geol. Geophys., 2009, no. 8, pp. 695–713.
Sokol, E.B., Maksimova, N.V., Nigmatulina, E.N., et al., Pirometamorfizm (Pyrometamorphism), Novosibirsk: SO RAN, 2005.
Sokol, E.V., Novikova, S.A., Alekseev, D.V., and Travin, A.V., Natural coal fires in the Kuznetsk coal basin: geologic causes, climate, and age, Russ. Geol. Geophys., 2014, vol. 55, no. 9, pp. 1043–1064.
Sokol, E., Sharygin, V., Kalugin, V., et al., Fayalite and kirschsteinite solid solutions in melts from burned spoilheaps, South Urals, Russia, Eur. J. Mineral., 2002, vol. 14, pp. 795–807.
Steiger, R.H. and Jäger, E., Subcommission on geochronology: convention on the use of decay constants in geoand cosmochronology, Earth Planet. Sci. Lett., 1977, vol. 36, pp. 359–362.
Wyllie, P.J. and Tuttle, O.F., The system CaO–CO2-H2O and the origin of carbonatites, J. Petrol., 1960, vol. 1, pp. 1–46.
Žacek, V., Skála, R., Chlupácová, M., and Dvorak, Z., Ca- Fe3+-rich, Si-undersaturated buchite from Želénky, North- Bohemian brown coal basin, Czech Republic, Eur. J. Mineral., 2005, vol. 17, pp. 623–633.
Žaček, V., Skála, R., and Zdeněk, D., Combustion metamorphism in the Most Basin, Coal and Peat Fires: A Global Perspective, Glenn, B., Prakash, A., and Sokol, E.V., Eds., New York: Elsevier, 2015, pp. 162–202.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.S. Peretyazhko, E.A. Savina, E.A. Khromova, N.S. Karmanov, A.V. Ivanov, 2018, published in Petrologiya, 2018, Vol. 26, No. 2, pp. 178–210.
Rights and permissions
About this article
Cite this article
Peretyazhko, I.S., Savina, E.A., Khromova, E.A. et al. Unique Clinkers and Paralavas from a New Nyalga Combustion Metamorphic Complex in Central Mongolia: Mineralogy, Geochemistry, and Genesis. Petrology 26, 181–211 (2018). https://doi.org/10.1134/S0869591118020054
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0869591118020054