Abstract
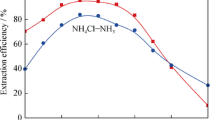
The extraction of zinc(II) ions with DX-510A β-diketone from sulfate and ammonia–sulfate solutions is reported. The effects of the aqueous phase pH and extracting agent concentration on the process have been investigated. The highest zinc(II) ion recovery is observed at pH 8.5–9.0. The time to extraction equilibrium is no longer than 30 s; in back extraction, it does not exceed 70 s. The extraction of zinc(II) ions is accompanied by the formation of an ammonia-containing organic complex. A chemical formula is suggested for this compound.
Similar content being viewed by others
References
Meng, X.H. and Kenneth, N.H., The principles and applications of ammonia leaching of metals–a review, Miner. Process. Extr. Metall. Rev., 1996, vol. 16, no. 1, pp. 23–61.
Dreisinger, D.B., Peters, E., Morgan, G., and Dreisinger, D.B., Hydrometallurgical treatments of carbon steel arc furnace dust, Hydrometallurgy, 1990, vol. 25, pp. 137–152.
Nyirenda, R.L. and Lugtmeijer, A.D., Ammonium carbonate leaching of carbon steelmaking dust: detoxification potential and economic feasibility of a conceptual process, Miner. Eng., 1993, vol. 6, no. 7, pp. 785–797.
Deep, A. and Carvalho J.M.R. Review on the recent developments in the solvent extraction of zinc, Solvent Extr. Ion Exch., 2008, vol. 26, no. 4, pp. 375–404.
Vahidi, E., Rashchi, F., and Moradkhani, D., Recovery of zinc from an industrial zinc leach residue by solvent extraction using D2EHPA, Miner. Eng., 2009, vol. 22, no. 2, pp. 204–206.
Alguacil, F.J. and Alonso, M., The effect of ammonium sulphate and ammonia on the liquid–liquid extraction of zinc using LIX 54, Hydrometallurgy, 1999, vol. 53, no. 2, pp. 203–209.
Rao, K.S., Sahoo, P.K., and Jena, P.K., Extractions of zinc from ammoniacal solutions by Hostarex DK-16, Hydrometallurgy, 1992, vol. 31, nos. 1–2, pp. 91–100.
Zhu, T, Extraction and Ion Exchange, Beijing Metall. Industry, 2005.
Kordosky, G.A., Virnig, M.J., and Mattison, P, β-Diketone copper extractants: Structure and stability, Cape Town: ISEC, 2002.
Gubin, A.F., Kolesnikov, V.A., Kondratyeva, E.S., and Il’in, V.I., Copper extraction from ammoniacal media, Khim. Prom–st. Segodnya, 2012, no. 6, pp. 36–42.
Piskareva, S.K., Barashkov, K.M., and Ol’shanova, K.M, Analiticheskaya khimiya (Analytical Chemistry), Moscow Vysshaya Shkola, 1994.
Kondratyeva, E.S., Gubin, A.F., and Kolesnikov, V.A., Nickel extraction from ammonia–sulfate solutions with the β-diketone DX-510A, Khim. Tekhnol., 2013, vol. 14, no. 12, pp. 745–751.
Zolotov, Yu.A, Ekstraktsiya vnutrikompleksnykh soedinenii (Extraction of Chelates), Moscow Nauka, 1968.
Nakamoto, K, Infrared and Raman Spectra of Inorganic and Coordination Compounds, New York Wiley, 1986.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.S. Kondratyeva, A.F. Gubin, V.A. Kolesnikov, 2016, published in Teoreticheskie Osnovy Khimicheskoi Tekhnologii, 2016, Vol. 50, No. 1, pp. 86–89.
Rights and permissions
About this article
Cite this article
Kondratyeva, E.S., Gubin, A.F. & Kolesnikov, V.A. Study of the extraction of zinc(II) ions from ammonia–sulfate solutions. Theor Found Chem Eng 50, 83–86 (2016). https://doi.org/10.1134/S0040579516010103
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0040579516010103