Abstract
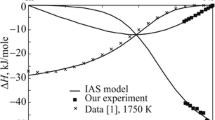
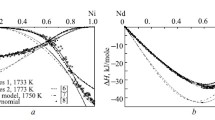
Enthalpies of mixing of liquid alloys are determined via isoperibol calorimetry for the Gd–Sn system throughout the range of concentrations at 1873, 1640, and 1510 K, and for the Gd–Sn–Ni system along the radial section with a constant ratio of atomic fractions of tin and nickel: xSn/xNi = 0.68/0.32 at 1873 ± 5 K up to xGd ∼ 0.25. It is found that the melts of the Gd–Sn system form with the release of a large amount of heat (\(\Delta {{{H}}_{{\min }}}\)= −68.4 ± 0.4 kJ/mol at xSn = 0.45). The calculations are performed using the geometric and analytical Redlich–Kister–Muggianu models of ΔH for melts of the ternary Gd–Sn–Ni system from similar data for binary boundary subsystems at 1873 K. The results show the minimum enthalpy of mixing of these ternary melts was established for the Gd0.55Sn0.45 alloy. It is shown that the experimentally investigated enthalpies of mixing of the Gd–Sn–Ni melts and calculated data using the Redlich–Kister–Muggianu model with ternary interaction parameter L = 500 kJ/mol agree satisfactorily with one another.









Similar content being viewed by others
REFERENCES
V. S. Sudavtsova, M. A. Shevchenko, M. I. Ivanov, V. V. Berezutskii, and V. G. Kudin, Russ. J. Phys. Chem. A 91, 990 (2017).
A. T. Dinsdale, CALPHAD 15, 319 (1991).
J. Kim and In-Ho Jung, CALPHAD 55, 134 (2016).
A. Percheron-Guegan, J. C. Achard, A. Bacha, et al., in Proceedings of the 10th Rare Earth Research Conference, Carefree, AZ, 1973 (U. S. Atomic Energy Commission Technical Information Center, Oak Ridge, TN, 1973), p. 1046; G. Borzone, A. Borsese, and R. Ferro, Z. Anorg. Allg. Chem. 601, 199 (1983).
N. G. Kulagina and A. P. Bayanov, Zh. Fiz. Khim. 48, 233 (1974).
Ling-Qi Liu and Jian-Xuan Zheng (C.S. Cheng), Acta Phys. Sin. 33, 1155 (1984).
N. G. Kulagina, A. P. Bayanov, and N. M. Kulagin, Izv. Akad. Nauk SSSR, Met., No. 3, 211 (1985).
V. N. Eremenko, M. V. Bulanova, and P. S. Martsenyuk, in Phase Equilibria, Structure, and Properties of Alloys (Nauk. Dumka, Kiev, 1990), p. 70 [in Russian].
A. Palenzona and S. Cirafici, J. Phase Equilib. 12, 690 (1991).
N. G. Kulagina, Extended Abstract of Cand. Sci. (Chem.) Dissertation (Sverdlovsk, 1980).
F. Sommer, J. Schott, and H.-G. Krull, J. Less-Common Met. 144, 53 (1988).
A. Bacha, C. Chatillon-Colinet, A. Percheron, and J.‑C. Mathieu, C. R. Acad. Sci. C 274, 680 (1972).
M. A. Shevchenko, M. I. Ivanov, V. V. Berezutskii, and V. S. Sudavtsova, Russ. J. Phys. Chem. A 90, 1 (2016).
C. Colinet, A. Pasturel, A. Percheron-Guegan, and J. C. Achard, J. Less-Common Met. 102, 167 (1984).
S. V. Meschel and O. J. Kleppa, J. Alloys Compd. 238, 180 (1996).
A. Palenzona, Thermochim. Acta 5, 473 (1973).
E. Bonnier and R. Caboz, C. R. Acad. Sci. (Paris) 250, 527 (1960).
G. W. Toop, Trans. Metall. Soc. AIME 233, 850 (1965).
F. Kohler and G. H. Findenegg, Monatsh. Chem. 96, 1228 (1965). https://doi.org/10.1007/BF00904272
Y. M. Muggianu, M. Gambino, and J. P. Bros, J. Chim. Phys. Phys.-Chim. Biol. 72, 83 (1975).
I. V. Nikolaenko and O. V. Vlasova, Rasplavy, No. 4, 12 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sudavtsova, V.S., Shevchenko, M.A., Kudin, V.G. et al. Thermodynamic Properties of Gd–Sn and Gd–Sn–Ni Melt Systems. Russ. J. Phys. Chem. 95, 237–243 (2021). https://doi.org/10.1134/S0036024421020254
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024421020254