Abstract
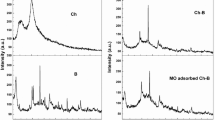
Trimeric surfactant modified montmorillonite immobilized in alginate beads (CA/3RenQ-Mt) sorbent was synthesized to simultaneously remove Cu2+ and methyl orange (MO) from aqueous solution. The CA/3RenQ-Mt was characterized using Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), thermogravimetric analysis (TG), zeta potential analysis and scanning electron microscopy/energy dispersive spectroscopy (SEM/EDS) analysis. The adsorption of Cu2+ and MO by CA/3RenQ-Mt was studied by the operational variables including contact time, pH and temperature. The results showed CA/3RenQ-Mt exhibited higher affinity to both Cu2+ and MO compared to sodium montmorillonite (Mt-Na), trimeric surfactant modified montmorillonite (3RenQ-Mt) or calcium alginate (CA). The pseudo-first-order kinetic model and Langmuir isotherm model were found to describe the MO adsorption well; the pseudo-second-order kinetic model and Freundlich isotherm model fitted well with the Cu2+ adsorption. The adsorption of MO onto CA/3RenQ-Mt is an endothermic spontaneous process, and ion-exchange was the dominant adsorption mechanism. Accordingly, the adsorption of Cu2+ on CA/3RenQ-Mt involved endothermic spontaneous chemical adsorption by means of coordination/chelation between Cu2+ and the oxygen atoms of carboxyl and hydroxyl groups of alginate. In general, CA/3RenQ-Mt can be applied as multifunctional adsorbent to remove heavy ions and anion dyes from aqueous solution.






Similar content being viewed by others
REFERENCES
K. U. Mohammad, Chem. Eng. J. 308, 438 (2017).
E. Abdelhahi, B. Michel, O. S. Mohamed, et al., Chem. Eng. J. 178, 168 (2011).
S. Ahmatmet, S. Gungor, T. Mustafa, Ind. Eng. Chem. Res. 51, 6877 (2012).
H. D. Mohammad, S. Daryoush, A. Imran, et al., J. Mol. Liq. 215, 671 (2016).
R. Vincent, B. Agnes, S. Jean-Michel, et al., J. Hazard. Mater. 178, 434 (2010).
O. H. Kwon, J. O. Kim, D. W. Cho, et al., Chemosphere 160, 126 (2016).
W. H. Luo, H. Tsuyoshi, and S. Keiko, Appl. Surf. Sci. 123, 29 (2016).
G. H. Xue, M. L. Gao, Z. Gu, et al., Chem. Eng. J. 218, 223 (2013).
C. Chen, H. B. Liu, T. H. Chen, et al., Appl. Clay Sci. 118, 239 (2015).
S. Zhang, J. Yang, X. Xin, et al., Environ. Prog. Sustain. 34, 39 (2015).
Y. G. Wang, L. H. Hua, G. Y. Zhang, et al., Colloid Interface Sci. 494, 380 (2017).
F. Wang, X. W. Lu, and X. Y. Li, J. Hazard. Mater. 308, 75 (2016).
S. H. Lin and R. S. Juang, J. Hazard. Mater. B 92, 315 (2002).
L. Y. Ma, Q. Z. Chen, J. X. Zhu, et al., Chem. Eng. J. 283, 880 (2016).
B. J. Hua and H. J. Luo, Appl. Surf. Sci. 257, 769 (2010).
V. V. Goncharuk, L. N. Puzyrnaya, G. N. Pshinko, et al., J. Water Chem. Technol. 33, 147 (2011).
X. B. Xing, P. H. Chang, G. C. Lv, et al., J. Taiwan Inst. Chem. E 59, 237 (2016).
Y. Q. Liang and H Li, J. Mol. Liq. 227, 139 (2017).
V. D. Maria, M. L. Maria, and S. D. Ecaterina, React. Funct. Polym. 93, 77 (2015).
T. K. Havva, Int. J. Biol. Macromol. 94, 202 (2017).
Y. F. Pan, P. X. Cai, F. F. Madjid, et al., Appl. Surf. Sci. 385, 333 (2016). http://www.sciencedirect.com/science/article/pii/S016943321631131X.
S. P. Santoso, L. Laysra, J. N. Putro, et al., J. Mol. Liq. 233, 29 (2017).
L. Aguero, Z. S. Dionisio, P. Luis, et al., Carbohydr. Polym. 168, 32 (2017).
S. C. Jang, S. M. Kang, Y. Haldorai, et al., Sci. Rep. 6, 38384 (2016).
S. K. Papageorgiou, F. K. Katsaros, E. P. Kouvelos, et al., J. Hazard. Mater. 137, 1765 (2006).
D. Nassima, B. Mokhtar, C. Nacer-Eddine, et al., Int. J. Biol. Macromol. 92, 1277 (2016).
T. Lu, T. Xiang, X. L. Huang, et al., Carbohyd. Polym. 133, 587 (2015).
T. Yoshimura, H. Yoshida, A. Ohno, et al., J. Colloid Interface Sci. 267, 167 (2003).
S. F. Lim, Y. M. Zheng, S. W. Zou, et al., Environ. Sci. Technol. 42, 2551 (2008).
X. Sun, J. H. Chen, Z. B. Su, et al., Chem. Eng. J. 211–212, 1 (2016).
M. Rola and C. Detellier, Materials 6, 5199 (2013).
Z. Q. Wang, Y. G. Huang, M. Wang, et al., J. Environ. Chem. Eng. 4, 3185 (2016).
Z. Qi, Q. Gao, W. J. Luo, et al., Colloid. Surf. A 470, 248 (2015).
Q. S. Zhou, X. Y. Lin, B. Li, et al., Chem. Eng. J. 256, 306 (2014).
ACKNOWLEDGMENTS
This work was supported by the Chinese Ministry of Education through a National Undergraduate Training Programs for Innovation and Entrepreneurship (201610122004), the Natural Science Foundation of Shanxi Province (201701D121034), the Fund for Shanxi Key Subjects Construction, and Support Plan for Innovative Research Team of Changzhi University, Science and Technology Innovation Doctoral Research Fund of Shanxi Agricultural University (no. 2015ZZ03).
Author information
Authors and Affiliations
Corresponding authors
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Li, H., Zhang, S.P., Liang, Y.Q. et al. Trimeric Surfactant Modified Montmorillonite Immobilized in Alginate Beads: An Efficient Adsorbent for Removal of Cu2+ and Methyl Orange from Aqueous Solution. Russ. J. Phys. Chem. 92, 2802–2810 (2018). https://doi.org/10.1134/S0036024418130186
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024418130186