Abstract
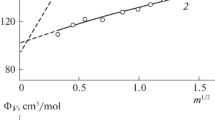
The heat capacity and bulk properties of barium and cadmium iodide solutions in dimethylformamide (DMF) at 298.15 K are measured via calorimetry and densimetry. Standard partial molar heat capacities \(\bar {C}_{{p,2}}^{^\circ }\) and volumes \(\bar {V}_{2}^{^\circ }\) ВаI2 and CdI2 in DMF are calculated. The standard values of heat capacity \(\bar {C}_{{p,i}}^{^\circ }\) and volume \(\bar {V}_{i}^{^\circ }\) of barium and cadmium in DMF at 298.15 K were determined.

Similar content being viewed by others
REFERENCES
A. N. Novikov and V. A. Vasilev, Russ. J. Phys. Chem. A 82, 1071 (2008).
A. N. Novikov, Russ. J. Phys. Chem. A 83, 334 (2009).
A. N. Novikov and L. Yu. Rassokhina, Russ. J. Phys. Chem. A 87, 1318 (2013).
L. Yu. Rassokhina and A. N. Novikov, Russ. J. Phys. Chem. A 89, 2226 (2015).
A. N. Novikov, Ya. I. Doronin, and P. A. Rakhmanova, Russ. J. Phys. Chem. A 92 (7), 1438 (2018).
B. V. Mikhailin, A. F. Vorob’ev, and V. A. Vasilev, Zh. Fiz. Khim. 56, 1937 (1982).
A. F. Kapustinskii, M. S. Stakhanova, and V. A. Vasilev, Izv. Akad. Nauk SSSR, Khim., No. 12, 2082 (1960).
Y. Marcus and G. Hefter, J. Chem. Soc., Faraday Trans. 92, 757 (1996).
Y. Marcus and G. Hefter, Chem. Rev. 104, 3427 (2004).
Y. Marcus, Ion Solvation (Wiley, Chichester, 1985).
L. P. Safonova, A. N. Kinchin, and A. M. Kolker, Russ. J. Phys. Chem. A 73, 1943 (1999).
R. Alexander and A. J. Parker, J. Am. Chem. Soc. 89, 5539 (1967).
M. R. Dack, K. J. Bird, and A. J. Parker, Aust. J. Chem. 28, 955 (1975).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by A. Bannov
Rights and permissions
About this article
Cite this article
Novikov, A.N., Doronin, Y.I., Vasilev, V.A. et al. Standard Partial Molar Heat Capacities and Volumes of Barium and Cadmium Ions in Dimethylformamide at 298.15 K. Russ. J. Phys. Chem. 92, 1641–1643 (2018). https://doi.org/10.1134/S0036024418080186
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024418080186