Abstract
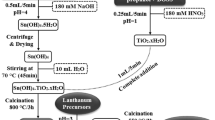
Surfactant-assisted synthesis of leucite (KAlSi2O6) nanoparticles was carried out by a hydrothermal method using an anionic surfactant at variable temperatures and surfactant concentrations. The newly synthesized leucite nanoparticles were characterized by FTIR, TGA, XRD, FESEM, and TEM. These nanoparticles have a wide and direct band gap at their smallest particle size (E g = 3.30 eV), showing a significant quantum confinement effect. Samples of leucite were prepared at 180°C with different SDS concentrations 0.006, 0.007, 0.008, 0.009, and 0.01 M and were used to degrade methylene blue under ultraviolet radiations. These samples degraded methylene blue to 18.5, 31.7, 45.81, 31.61, 30.1%, respectively. The most effective catalyst is the one which was synthesized at 200°C and the CMC value of the surfactant (sodium dodecyl sulfate) having the percentage degradation of 49.1%.
Similar content being viewed by others
References
P. H. Ribbe, Am. Mineral. 64, 402 (1979).
D. C. Palmer, M. T. Dove, R. M. Ibberson, et al., Am. Mineral. 82, 16 (1997).
O. Y. Golubeva, N. Y. Ul’yanova, and L. N. Kurilenko, Glass Phys. Chem. 39, 649 (2013).
T.-S. Sheu, W. J. O’Brien, S. T. Rasmussen, et al., J. Mater. Sci. 29, 125 (1994).
Y. Zhang, J. Wu, P. Rao, and M. Lv, Mater. Lett. 60, 2819 (2006).
M. Novotna, A. Kloužkova, J. Maixner, et al., Ceram. Silikaty 49, 252 (2005).
V. Šatava, A. Kloužkova, D. Ležal, et al., Ceram. Silikaty 46, 37 (2002).
A. Balandis and I. Sinkyavichene, Glass Ceram. 62, 49 (2005).
Y. Zhang, C. Qu, J. Wu, et al., J. Wuhan Univ. Technol.-Mat. Sci. Ed. 23, 452 (2008).
Y. Zhang, B. Li, P. Rao, et al., J. Am. Ceram. Soc. 90, 1615 (2007).
A. Imtiaz, M. A. Farrukh, M. Khaleeq-ur-Rahman, et al., Sci. World J. 2013, 641420 (2013).
R. Adnan, N. A. Razana, I. A. Rahman, et al., J. Chin. Chem. Soc. 57, 222 (2010).
K. M. A. Saron, M. R. Hashim, and M. A. Farrukh, Superlatt. Microstruct. 64, 88 (2013).
K. M. A. Saron, M. R. Hashim, and M. A. Farrukh, Appl. Surf. Sci. 258, 5200 (2012).
E. M. Mkawi, K. Ibrahim, M. K. M. Ali, et al., Appl. Mech. Mater. 343, 85 (2013).
R. L. Ziolli and W. F. Jardim, Quimica Nova 21, 319 (1998).
T. N. M. Cervantes, D. A. M. Zaia, and H. de Santana, Quimica Nova 32, 2423 (2009).
J. P. Tardivo, A. del Giglio, C. S. de Oliveira, et al., Photodiagn. Photodyn. Ther. 2, 175 (2005).
H. Yao, N. Li, S. Xu, et al., Biosens. Bioelectron. 21, 372 (2005).
M. Arvand, S. Sohrabnezhad, M. F. Mousavi, et al., Anal. Chim. Acta 491, 193 (2003).
K. P. Singh, D. Mohan, S. Sinha, et al., Ind. Eng. Chem. Res. 42, 1965 (2003).
P. Nigam, G. Armour, I. M. Banat, et al., Biores. Technol. 72, 219 (2000).
A. S. Özcan and A. Özcan, J. Colloid Interface Sci. 276, 39 (2004).
A. Gürses, S. Karaca, Ç. Doğar, et al., J. Colloid Interface Sci. 269, 310 (2004).
G. Anderson and C. W. Burnham, Am. J. Sci. 283, 283 (1983).
A. Gaber, A. Y. Abdel-Latief, M. A. Abdel-Rahim, et al., Mater. Sci. Semicon. Proc. 16, 1784 (2013).
A. Thottoli and A. Unni, J. Nanostruct. Chem. 3, 1 (2013).
E. M. Mkawi, K. Ibrahim, M. K. M. Ali, et al., J. Mater. Sci.: Mater. Electron. 25, 857 (2014).
Y. Wang and N. Herron, J. Phys. Chem. 95, 525 (1991).
K. Kabra, R. Chaudhary, and R. L. Sawhney, Ind. Eng. Chem. Res. 43, 7683 (2004).
G. Huey-Shya, R. Adnan, and M. A. Farrukh, Turk. J. Chem. 35, 375 (2011).
M. Shahid, M. A. Farrukh, A. A. Umar, et al., Russ. J. Phys. Chem. A 88, 842 (2014).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Farrukh, M.A., Naseem, F., Imtiaz, A. et al. Hydrothermal synthesis of leucite nanoparticles using anionic surfactant: Structural evaluation and catalytic properties. Russ. J. Phys. Chem. 90, 1231–1237 (2016). https://doi.org/10.1134/S0036024416060145
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024416060145