Abstract
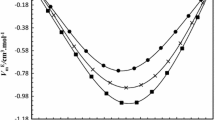
The curve of the concentration dependence of the total molecular light scattering coefficient of xC3H7OH-(1 − x)H2O solutions showed two sharp maxima (at x = 0.04 and 0.09) and a minimum at x = 0.06 mole fractions, suggesting a considerable rearrangement of the structure of the solutions near these concentrations. The concentration dependences of excess molar volume, adiabatic compressibility, and molar refraction showed negative deviations from the ideal with a minimum in the range x = 0.18–0.3. The structure of water was assumed to undergo a rearrangement at x = 0.04 accompanied by structural fluctuations, which dispersed at 0.04 < x < 0.06. At x = 0.06, associates of isopropanol molecules started to form, whose number and size increased linearly up to x = 0.13. In the 0.13 < x < 0.35 concentration range, some kind of an aqueous alcohol emulsion containing pure alcohol “nanodrops” formed and the solutions became microheterogeneous.
Similar content being viewed by others
References
I. S. Ryzhkina, Yu. V. Kiseleva, G. A. Zheltukhina, S. A. Okorochenkov, L. I. Murtazina, A. P. Timosheva, V. E. Nebolsin, and A. I. Konovalov, Dokl. Ross. Phys. Chem. 440, 157 (2011).
M. E. Panov and O. B. Sokolova, Russ. J. Gen. Chem. 73, 93 (2003).
M. Sedlak, J. Phys. Chem. B 117, 2495 (2013).
R. S. Anderson and M. C. R. Symons, Trans. Faraday Soc. 65, 2550 (1969).
M. D’Angelo, G. Onori, and A. Santucci, J. Chem. Phys. 100, 3107 (1994).
M. I. Davis and E. S. Ham, Thermochim. Acta 190, 251 (1991).
S. I. Sivakova, E. Yu. Korableva, and L. V. Lanshina, Russ. J. Phys. Chem. A 69, 913 (1995).
M. F. Vuks and L. V. Shurupova, Opt. Commum. 5, 273 (1972).
M. F. Vuks, Light Scattering in Gases, Liquids, and Solutions (Leningr. Gos. Univ., Leningrad, 1977) [in Russian].
H. Wahid, J. Mol. Liquids 55, 73 (1993).
L. V. Lanshina, Russ. J. Phys. Chem. A 72, 1110 (1998).
G. I. Egorov, V. N. Afanas’ev, and A. M. Kolker, Russ. J. Gen. Chem. 74, 171 (2004).
V. N. Afanas’ev, L. S. Efremova, and T. V. Volkova, in Physicochemical Properties of Binary Solvents. Aqueous Systems (Inst. Khimii Nevodnykh Rastvorov AN SSSR, Ivanovo, 1988), Ch. 2 [in Russian].
A. I. Abramovich and L. V. Lanshina, Russ. J. Phys. Chem. A 88, 807 (2014).
L. V. Lanshina and A. I. Abramovich, Russ. J. Phys. Chem. A 79, 608 (2005).
G. Onori, J. Chem. Phys. 89, 1251 (1998).
K. Jerie, A. Baranowski, and B. Rosenfeld, Phys. Scripta 35, 729 (1987).
A. Baranowski, K. Jerie, and I. Glinski, Chem. Phys. 214, 143 (1997).
H. Endo, Bull. Chem. Soc. Jpn. 46, 3004 (1973).
L. V. Lanshina, E. Yu. Korableva, and V. I. Zhuravlev, in Proceedings of the 4th International Conference on Dielectric and Related Phenomena, Poland, September 16–20, 1996, p. 52.
P. A. Khakimov, G. M. Dakar, A. K. Karshivaev, et al., Uzb. Fiz. Zh., No. 1, 94 (1992).
L. V. Lanshina, M. N. Rodnikova, and K. T. Dudnikova, Zh. Strukt. Khim. 31, 65 (1990).
L. V. Lanshina, M. N. Rodnikova, and K. T. Dudnikova, Vestn. Mosk. Univ., Ser. 2: Khim. 30, 111 (1989).
T. Ramanjappa and E. Rajagopiab, Bull. Chem. Soc. Jpn. 61, 1271 (1988).
L. Jaime and J. E. Desnoyers, J. Solution Chem. 10, 465 (1981).
M. J. Blandamer, N. J. Hidden, M. C. R. Symons, et al., Trans. Faraday Soc. 64, 66 (1967).
R. F. Lama and C. Y. Benjamin, J. Chem. Eng. Data 10, 216 (1965).
G. Roux, D. Roberts, G. Perron, et al., J. Solution Chem. 9, 629 (1980).
A. Nilson, Chem. Phys. Lett. 460, 387 (2008).
L. Ralph, Chem. Phys. Chem. 8, 938 (2007).
J. Marcus, J. Mol. Liq. 166, 62 (2012).
V. M. Byakov, L. V. Lanshina, A. P. Stepanova, and S. V. Stepanov, Russ. J. Phys. Chem. A 83, 214 (2009).
Fan Jin, Ye Jing, L. Hong, et al., J. Phys. Chem. B 111, 2255 (2007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.I. Abramovich, L.V. Lanshina, 2015, published in Zhurnal Fizicheskoi Khimii, 2015, Vol. 89, No. 7, pp. 1116–1121.
Rights and permissions
About this article
Cite this article
Abramovich, A.I., Lanshina, L.V. Analysis of the structure of aqueous solutions of isopropanol based on optical, volumetric, and elastic data. Russ. J. Phys. Chem. 89, 1211–1215 (2015). https://doi.org/10.1134/S003602441507002X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602441507002X