Abstract
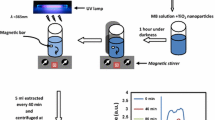
TiO2 and Sn/Cu-TiO2 nanoparticles with different Sn/Cu contents have been synthesized and characterized by different analysis methods such as XRD, TEM, and BET. Sn/Cu-TiO2 was preferential to TiO2 with 2,5-fold increase in photodegradation rate of different types of dyes such as methyl orange, methylene blue and methyl red. This activity was relevant to the influences of co-doping on the physicochemical properties and surface interfacial charge transfer mechanisms. An optimum synergetic effect was found for a mass ratio of Sn/TiO2 equal to 0.75% and Cu/TiO2 equal to 0.5%. The degradation reaction fit well to a Langmuir-Hinshelwood kinetic model indicating the reaction rate is depended on initial adsorption step.
Similar content being viewed by others
References
M. Zhou, J. Yu, and B. Cheng, J. Hazard. Mater. 137, 1838 (2006).
O. Legrini, E. Oliveros, and A. M. Braun, Chem. Rev. 93, 671 (1993).
T. Sugimoto, X. Zhou, and A. Muramatsu, Colloid Interface Sci. 252, 347 (2003).
W. Y. Choi, A. Termin, and M. R. Hoffmann, Phys. Chem. 98, 13669 (1994).
C. Chen, Z. Wang, S. Ruan, B. Zou, M. Zhao, and F. Wu, Dyes Pigments 77, 204 (2008).
G. Sivalingam, K. Nagaveni, M. S. Hegde, and G. Madras, Appl. Catal. B 45, 23 (2003).
C. X. Hu, Y. S. Wu, N. Lun, and Y. C. Shi, Mater. Sci. Eng. B 110, 1 (2004).
M. A. Saepurahman, Abdullah, and F. K. Chong, J. Hazard. Mater. 176, 451 (2010).
J. M. Herrmann, Catal. Today. 53, 115 (1999).
N. R. C. Fernandes Machado and V. S. Santana, Catal. Today 107, 595 (2005).
I. K. Konstantinou and T. A. Albanis, Appl. Catal. B 49, 1 (2004).
Y. Bessekhouad, D. Robert, and J. V. Weber, J. Photochem. Photobiol. A: Chem. 157, 47 (2003).
M. A. Behnajady, H. Eskandarloo, N. Modirshahla, and M. Shokri, Desalination 278, 10 (2011).
M. Hamadanian, A. Reisi-Vanani, and A. Majedi, Iran. Chem. Soc. 7, S52 (2010).
R. A. Spurr and H. Myers, Anal. Chem. 29, 760 (1957).
B. Tryba, M. Piszcz, B. Grzmil, A. Pattek-Janczyk, and A. W. Morawski, Hazard. Mater. 162, 111 (2009).
C. R. Estrellan, C. Salim, and H. Hinode, J. Hazard. Mater. 179, 79 (2010).
J. G. Yu, J. C. Yu, and W. K. Ho, Appl. Catal. A: Gen. 255, 309 (2003).
S. Rahimnejad, S. R. Setayesh, and M. R. Gholami, J. Iran. Chem. Soc. 5, 367 (2008).
L. Kumaresan, M. Mahalakshmi, M. Palanichamy, and V. Murugesan, Indust. Eng. Chem. Res. 49, 1480 (2010).
X. Yang, C. Cao, L. Erickson, K. Hohn, R. Maghirang, and K. Klabunde, Appl. Catal. B: Environ. 91, 657 (2009).
G. Colon, M. Maicu, M. C. Hidalgo, and J. A. Navio, Appl. Catal. B: Environ. 67, 41 (2006).
Y. Kotani, T. Matoda, A. Marsuda, T. Kogure, and M. Tatsumisago, and T. Minami, Mater. Chem. 11, 2045 (2001).
J. Li, J. Xu, W. L. Dai, H. Li, and K. Fan, Appl. Catal. B 85, 162 (2009).
N. Venkatachalam, M. Palanichamy, B. Arabindoo, and V. Murugesan, J. Catal. Commun. 8, 1088 (2007).
S. S. Srinivasan, J. Wade, E. K. Stefanakos, and Y. Goswami, Alloys Compd. 424, 322 (2006).
J. G. Yu, X. J. Zhao, J. C. Du, and W. M. Chen, Sol-Gel Sci. Tech. 17, 163 (2000).
M. Zhou, J. Yu, and B. Cheng, J. Hazard. Mater. 137, 1838 (2006).
O. Carp, C. L. Huisman, and A. Reller, Progress Solid State Chem. 32, 33 (2004).
H. M. Coleman, K. Chiang, and R. Amal, Chem. Eng. J. 113, 65 (2005).
N. Venkatachalam, M. Palanichamy, B. Arabindoo, and V. Murugesan, J. Catal. Commun. 8, 1088 (2007).
B. H. Hameed, R. R. Krishni, and S. A. Sata, J. Hazard. Mater. 162, 305 (2009).
C. Galindo, P. Jacques, and A. Kalt, Chemosphere 45, 997 (2001).
N. Daneshvar, D. Salari, and A. R. Khataee, J. Photochem. Photobiol. A: Chem. 157, 111 (2003).
N. Barka, A. Assabbane, A. Nounah, and Y. A. ît Ichou, Hazard. Mater. 152, 1054 (2008).
M. A. Behnajady, H. Eskandarloo, N. Modirshahla, and M. Shokri, Dig. J. Nanomater. Biostruct. 6, 1887 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Mohammadi, R., Massoumi, B. Sn/Cu-TiO2 nanoparticles produced via sol-gel method: Synthesis, characterization, and photocatalytic activity. Russ. J. Phys. Chem. 88, 1184–1190 (2014). https://doi.org/10.1134/S0036024414070243
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024414070243