Abstract
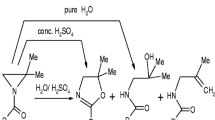
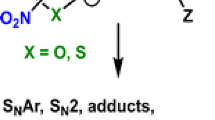
The alkylation reactions of the ambident ethyl acetoacetate anion with C2H5X (X = F, Cl, Br, and I) in the O2, C3, and O4 positions of the anion were investigated at the B3LYP/6-311+G(d,p) level of theory. It was found that the ethylation reaction does not occur in the position O4, as well as with ethyl fluoride in any position of the anion, due to very high activation energies and thermodynamic instability of the hypothetic products. The activation energies for the reactions in the position O2 are lower in comparison to the position C3, but the products of the reactions in the C3 position are more stable than those in the position O4, implying that the C/O products ratio is controlled by both thermodynamic and kinetic factors, leading to the O2-product with the chloride, and C3-product with the iodide as leaving group.
Similar content being viewed by others
References
L. Claisen and A. Claparede, Ber. Dtsch. Chem. Ges. 14, 2460 (1881).
L. Claisen, Ber. Dtsch. Chem. Ges. 20, 655 (1887).
J. B. Cohen, Practical Organic Chemistry for Advanced Students (MacMillan, London, 1907).
F. A. Carey, Organic Chemistry, 6th ed. (McGraw-Hill, New York, 2006).
B. Mehta and M. Mehta, Organic Chemistry (PHI Learning, New Delhi, 2005).
R. A. Ford and C. Letizia, Food Chem Toxicol. 26, 315 (1988).
M. Baumann, I. R. Baxendale, S. V. Ley, and N. Nikbin, Beilstein J. Org. Chem. 7, 442 (2011).
T. B. Johnson and D. A. Hahn, Chem. Rev. 13, 193 (1933).
E. R. Buchman, J. Am. Chem. Soc. 58, 1803 (1936).
M. Khazaei, M. A. Abbasinejad, A. Hassanabadi, and B. Sadeghi, E-J. Chem. 9, 615 (2012).
A. L. Kurts, N. K. Genkina, A. Macias et al., Tetrahedron 27, 4777 (1971).
T._L. Ho, Hard and Soft Acids and Bases Principle in Organic Chemistry (Academic Press, New York, 1977).
R. G. Pearson and J. Songstad, J. Am. Chem. Soc. 89, 1827 (1967).
W. J. Hehre, A. J. Shusterman, and J. E. Nelson, The Molecular Modeling Workbook for Organic Chemistry (Wavefunction, Irvine, 1998).
F. A. Carey and R. J. Sundberg, Advanced Organic Chemistry (Plenum Press, New York, 2007) pp. 253–388.
M. J. Frisch, G. W. Trucks, H. B. Schlegel et al., Gaussian 09 (Gaussian Inc., Wallingford CT, 2009).
C. Lee, W. Yang, and R. G. Parr, Phys. Rev. B 37, 785 (1988).
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
C. Peng, P. Y. Ayala, H. B. Schlegel, and M. J. Frisch, J. Comput. Chem. 17, 49 (1996).
C. Gonzalez and H. B. Schlegel, J. Chem. Phys. 90, 2154 (1989).
W. Koch and M. C. Holthausen, A Chemist’s Guide to Density Functional Theory (Wiley-VCH, Weinheim, 2001).
W. J. Hehre, A Guide to Molecular Mechanics and Quantum Chemical Calculations (Wavefunction Inc., Irvine, 2003).
S. N. Bachrach, Computational Organic Chemistry (Wiley, Hoboken, New Jersey, 2007).
W. Kohn, A. D. Becke, and R. G. Parr, J. Phys. Chem. 100, 12974 (1996).
E. J. Baerends, Theor. Chem. Acc. 103, 265 (2000).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Marković, S., Đurđević, J., Vukosavljević, M. et al. Mechanistic insight into alkylation of the ethyl acetoacetate anion with different ethyl halides. Russ. J. Phys. Chem. 87, 2207–2213 (2013). https://doi.org/10.1134/S0036024413130165
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024413130165