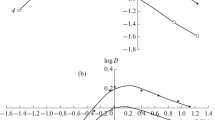
Abstract
It was found that the size of quaternary ammonium salt cations strongly influenced the log\( K_{Cl^ - }^{NO_3^ - } \) value, which was explained by the special features of ionic association in the organic phase. The use of 3,4,5-tridodecyloxybenzyltridodecylammonium nitrate as an electrode active substance could increase the selectivity of the NO −3 -selective electrode with respect to Cl− ions.
Similar content being viewed by others
References
E. M. Rakhman’ko, N. A. Sloboda, and O. A. Trosko, Zh. Neorg. Khim. 37, 1652 (1992).
I. V. Kachanovich, Tew Van Bien, and E. M. Rakhman’ko, Zh. Neorg. Khim. 39,347 (1994).
V. V. Egorov, N. D. Borisenko, E. M. Rakhman’ko, et al., Talanta 44, 1735 (1997).
E. M. Rakhman’ko, V. V. Egorov, E. B. Okaev, and E. V. Pomelenok, Dokl. Nats. Akad. Nauk Belarusi 47, 50 (2003).
V. V. Egorov, E. M. Rakhman’ko, and E. V. Pomelenok, Vestn. Bel. Gos. Univ., No. 2, 3 (2004).
E. V. Pomelenok, Candidate’s Dissertations in Chemistry (Bel. Gos. Univ., Minsk, 2004), p. 138.
N. A. Izmailov, Solution Electrochemistry (Mir, Moscow, 1986) [in Russian].
E. B. Okaev, Vestsi NAN Belarusi, Ser. Khim., No. 1, 52 (2005).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.M. Rakhman’ko, M.S. Markovskaya, L.S. Stanishevskii, Yu.S. Zubenko, A.R. Tsyganov, 2009, published in Zhurnal Fizicheskoi Khimii, 2009, Vol. 83, No. 10, pp. 1980–1985.
Rights and permissions
About this article
Cite this article
Rakhman’ko, E.M., Markovskaya, M.S., Stanishevskii, L.S. et al. The influence of the structure of the higher quaternary ammonium salt cation on chloride-nitrate exchange. Russ. J. Phys. Chem. 83, 1794–1799 (2009). https://doi.org/10.1134/S003602440910029X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602440910029X