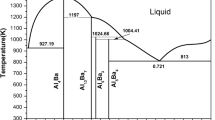
Abstract
The paper presents the results of the thermodynamic modeling of the composition and state of aggregation of the condensed medium formed in a Bi2O3-Ar medium at 9.8066 × 10−2 MPa over the temperature range 300–1300 K. The possible presence of the Bi2O3, BiO, Bi2O5, and BiO2 oxides in the solid and liquid states was taken into account. Temperature-induced changes in the solid and liquid solution components (bismuth oxides in various states of aggregation) are discussed.
Similar content being viewed by others
References
Concise Chemical Encyclopedia, Ed. by I. L. Knunyants (Sovetskaya Entsiklopediya, Moscow, 1961), Vol. 1 [in Russian].
I. A. Toropov, V. P. Barzakovskii, I. A. Bondar’, and Yu. P. Udalov, State Diagrams of Silicate Systems (Nauka, Leningrad, 1970) [in Russian].
R. Ripan and I. Ceteanu, Chimia metalelor (Bucuresti: Editura didactica si pedagogica, 1970; Mir, Moscow, 1972).
Physico-Chemical Properties of Oxides, Ed. by G. V. Samsonov (Metallurgiya, Moscow, 1978) [in Russian].
N. A. Vatolin, G. K. Moiseev, and B. G. Trusov, Thermodynamic Modeling in High-Temperature Inorganic Systems (Metallurgiya, Moscow, 1994) [in Russian].
G. K. Moiseev, N. A. Vatolin, and N. V. Belousova, Zh. Fiz. Khim. 74(12), 2138 (2000) [Russ. J. Phys. Chem. 74 (12), (2000)].
G. K. Moiseev, N. A. Vatolin, L. A. Marshuk, and N. I. Il’inykh, Temperature Dependences of Reduced Gibbs Energy for Some Inorganic Substances (ASTRA. OWN Alternative Databank) (Izd. Ural’sk. Otd. Ross. Akad. Nauk, Yekaterinburg, 1997) [in Russian].
G. K. Moiseev and N. A. Vatolin, in “IV Bilateral Russian-German Symposium “Physics and Chemistry of Novel Materials” (Inst. SOlid State Chemistry, Yekaterinburg, 1999).
G. K. Moiseev, N. A. Vatolin, and N. V. Belousova, Zh. Fiz. Khim. 74(7), 1180 (2000) [Russ. J. Phys. Chem. 74 (7), 1054 (2000)].
G. K. Moiseev, N. A. Vatolin, and N. V. Belousova, Zh. Fiz. Khim. 74(9), 1570 (2000) [Russ. J. Phys. Chem. 74 (9), 1415 (2000)].
V. M. Denisov, N. V. Belousova, G. K. Moiseev, et al., Mismuth-Containing Materials: Structure and Chemical Properties (Izd. Ural’sk. Otd. Ross. Akad. Nauk, Yekaterinburg, 2000) [in Russian].
Yu. F. Kargin, Zh. Neorg. Khim. 38(10), 1639 (1993).
K. N. Marushkin and A. S. Alikhanyan, Dokl. Akad. Nauk 329(4), 452 (1993).
A. N. Romanov, D. P. Shashkin, and E. V. Khaula, Zh. Neorg. Khim. 45(4), 570 (2000) [Russ. J. Inorg. Chem. 45 (4), 570 (2000)].
R. A. Lidin, V. A. Molochko, and L. L. Andreeva, Chemical Properties of Inorganic Substances (Nauka, Moscow, 1997) [in Russian].
G. K. Moiseev and A. L. Ivanovskii, Electronic Journal “Issledovano v Rossii,” 1544 (2005); http://zhurnal.ape.relarn.ru/artisles/2005/147.pdf.
Ya. A. Ugai, General and Inorganic Chemistry (Vysshaya Shkola, Moscow, 1997) [in Russian].
Properties of Elements: A Handbook, Ed. by M. A. Drits (Moscow, 1997), Vols. 1–2 [in Russian].
G. K. Moiseev and G. P. Vyatkin, Thermodynamic Modeling in Inorganic Systems (Izd. Yuzhn.-Ural’sk. Gos. Univ., Chelyabinsk, 1999) [in Russian].
G. B. Sinyarev, N. A. Vatolin, B. G. Trusov, and G. K. Moiseev, Application of Computers for Thermodynamic Calculations of Metallurgical Processes (Nauka, Moscow, 1982) [in Russian].
G. K. Moiseev and B. G. Trusov, Thermodynamic Modeling in High-Temperature Inorganic Systems (Izd. Ural’sk. Gos. Univ., Sverdlovsk, 1989) [in Russian].
G. K. Moiseev and N. A. Vatolin, J. Therm. Anal. 48, 1069 (1997).
G. K. Moiseev and N. A. Vatolin, Zh. Fiz. Khim. 71(3), 399 (1997) [Russ. J. Phys. Chem. 71 (3), 335 (1997)].
G. K. Moiseev and N. A. Vatolin, Zh. Fiz. Khim. 72(9), 1554 (1998) [Russ. J. Phys. Chem. 72 (9), 1398 (1998)].
G. K. Moiseev and N. A. Vatolin, Zh. Fiz. Khim. 72(9), 1559 (1998) [Russ. J. Phys. Chem. 72 (9), 1403 (1998)].
Author information
Authors and Affiliations
Additional information
Original Russian Text © G.K. Moiseev, A.L. Ivanovskii, 2006, published in Zhurnal Fizicheskoi Khimii, 2006, Vol. 80, No. 12, pp. 2123–2126.
Rights and permissions
About this article
Cite this article
Moiseev, G.K., Ivanovskii, A.L. The numerical modeling of the composition and state of aggregation of the condensed medium formed in heating the Bi2O3 oxide in argon. Russ. J. Phys. Chem. 80, 1887–1890 (2006). https://doi.org/10.1134/S0036024406120028
Received:
Issue Date:
DOI: https://doi.org/10.1134/S0036024406120028