Abstract
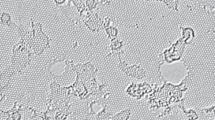
Film samples produced by codeposition of salts Na(form), Ni(form)2, Co(Аcac)3, and Pb(Аc)2 from graphene oxide dispersions are studied. The thus-prepared materials are characterized by spectral and X-ray analytical methods. The structure of graphene oxide containing salt crystals is studied by scanning electron microscopy (SEM). Crystallization occurs both on the surface and in the interlayer spaces of graphene oxide. No side processes occur during crystallization; the precursor salts retain their phase composition, except for lead acetate, which is partially hydrolyzed in aqueous solution. The incorporation of salt crystals increases film imperfection and enhances the resistivity of the material severalfold. The results of this study can appear useful in the development of graphene oxide based membranes.







Similar content being viewed by others
REFERENCES
I. V. Kozerozhets, G. P. Panasyuk, E. A. Semenov, et al., Ceram. Int. 48, 7522 (2022). https://doi.org/10.1016/j.ceramint.2021.11.296
I. V. Kozerozhets, G. P. Panasyuk, E. A. Semenov, et al., Ceram. Int. 46, 28961 (2020). https://doi.org/10.1016/j.ceramint.2020.08.067
G. P. Panasyuk, I. V. Kozerozhets, E. A. Semenov, et al., Inorg. Mater. 55, 920 (2019). https://doi.org/10.1134/S0020168519090127
G. P. Panasyuk, L. A. Azarova, V. N. Belan, et al., Theor. Found. Chem. Eng. 52, 879 (2018). https://doi.org/10.1134/S0040579518050202
A. A. Mikhaylov and A. G. Medvedev, Mason, et al., J. Mater. Chem. A 3, 20681 (2015). https://doi.org/10.1039/c5ta04514b
Y. V. Ioni, S. V. Kraevsky, Y. A. Groshkova, et al., Mendeleev Commun. 31, 718 (2021). https://doi.org/10.1016/j.mencom.2021.09.042
Z. Xu, S. Zhu, M. Wang, et al., ACS Appl. Mater. Interfaces 7, 1355 (2015). https://doi.org/10.1021/am507798d
Z. U. Khan, A. Kausar, H. Ullah, et al., J. Plast. Film Sheeting 32, 336 (2016). https://doi.org/10.1177/8756087915614612
O. Mrózek, L. Melounková, D. Smržová, et al., J. Hazard. Mater. 398, 123114 (2020). https://doi.org/10.1016/j.jhazmat.2020.123114
S. Shahriari, M. Sastry, S. Panjikar, et al., Nanotechnol. Sci. Appl. 14, 197 (2021). https://doi.org/10.2147/NSA.S334487
Matharu R. Kaur, T. A. Tabish, T. Trakoolwilaiwan, et al., J. Colloid Interface Sci. 571, 239 (2020). https://doi.org/10.1016/j.jcis.2020.03.037
Y. V. Ioni, S. E. Lyubimov, V. A. Davankov, et al., Russ. J. Inorg. Chem. 58, 392 (2013). https://doi.org/10.1134/s0036023613040062
L. Fritea, F. Bănică, T. O. Costea, et al., J. Electroanal. Chem. 830–831, 63 (2018). https://doi.org/10.1016/j.jelechem.2018.10.015
K. Z. Riahi, N. Sdiri, D. J. Ennigrou, et al., J. Mol. Struct. 1216, 128304 (2020). https://doi.org/10.1016/j.molstruc.2020.128304
Y. V. Ioni, Y. A. Groshkova, S. P. Gubin, et al., Nanotechnol. Russ. 15, 163 (2020). https://doi.org/10.1134/S1995078020020111
D. Dikin, S. Stankovich, E. Zimney, et al., Nature 448, 457 (2007). https://doi.org/10.1038/nature06016
Y. V. Ioni, Y. A. Groshkova, E. Y. Buslaeva, et al., Russ. J. Inorg. Chem. 66, 950 (2021). https://doi.org/10.1134/S0036023621060115
D. I. Petukhov, O. O. Kapitanova, E. A. Eremina, et al., Mendeleev Commun. 31, 137 (2021). https://doi.org/10.1016/j.mencom.2021.03.001
W. Cha-Umpong, E. Hosseini, A. Razmjou, et al., J. Membr. Sci. 598, 117687 (2019). https://doi.org/10.1016/j.memsci.2019.117687
M. B. M. Y. Ang, M. R. Gallardo, G. V. C. Dizon, et al., J. Membr. Sci. 587, 117188 (2019). https://doi.org/10.1016/j.memsci.2019.117188
L. Chen, G. Shi, J. Shen, et al., Nature 550, 380 (2017). https://doi.org/10.1038/nature24044
J.-C. Li, P.-X. Hou, and C. Liu, Small 13, 1702002 (2017). https://doi.org/10.1002/smll.201702002
A. S. Kuzenkova, A. Y. Romanchuk, A. L. Trigub, et al., Carbon 158, 291 (2019). https://doi.org/10.1016/j.carbon.2019.10.003
M. Wang, Y. Niu, J. Zhou, et al., Nanoscale 14587 (2016). https://doi.org/10.1039/c6nr03503e
R. R. Amirov, J. Shayimova, Z. Nasirova, et al., Carbon 116, 356 (2017). https://doi.org/10.1016/j.carbon.2017.01.095
A. Osman, S. Leaper, V. Sreepal, et al., Cryst. Growth Des. 19, 498 (2018). https://doi.org/10.1021/acs.cgd.8b01597
I. V. Kozerozhets, G. P. Panasyuk, E. A. Semenov, et al., Inorg. Mater. 56, 716 (2020). https://doi.org/10.1134/S002016852007009
G. P. Panasyuk, I. V. Kozerozhets, I. L. Voroshilov, et al., Russ. J. Phys. Chem. 89, 592 (2015). https://doi.org/10.1134/S0036024415040196
G. P. Panasyuk, E. A. Semenov, I. V. Kozerozhets, et al., Dokl. Chem. 483, 272 (2018). https://doi.org/10.1134/S0012500818110022
G. P. Panasyuk, V. N. Belan, I. L. Voroshilov, et al., Inorg. Mater. 46, 747 (2010). https://doi.org/10.1134/S0020168510070113
I. V. Kozerozhets, G. P. Panasyuk, E. A. Semenov, et al., Russ. J. Inorg. Chem. 65, 1529 (2020). https://doi.org/10.1134/S0036023620100149
I. M. Karzov, N. E. Sorokina, and V. V. Avdeev, Inorg. Mater. 45, 374 (2009). https://doi.org/10.1134/s0020168509040086
M. I. Saidaminov, N. V. Maksimova, N. E. Sorokina, et al., Inorg. Mater. 49, 883 (2013). https://doi.org/10.1134/s0020168513090161
ACKNOWLEDGMENTS
Facilities of the Shared Facilities Center of the Kurnakov Institute of General and Inorganic Chemistry, the Russian Academy of Sciences were used in the X-ray diffraction studies. For scanning electron microscopy experiments, facilities of the Educational and Methodological Center for Lithography and Microscopy at Moscow State University were used.
Funding
The study was supported by the Russian Science Foundation (project No. 22-19-00110).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Ioni, Y.V., Chentsov, S.I., Sapkov, I.V. et al. Preparation and Characterization of Graphene Oxide Films with Metal Salts. Russ. J. Inorg. Chem. 67, 1711–1717 (2022). https://doi.org/10.1134/S0036023622601076
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023622601076