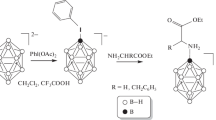
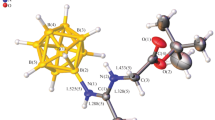
Abstract
A method is proposed for obtaining glycine-based closo-decaborate anion derivatives containing a fluorescent group to study the distribution of potential BNCT agents.

Similar content being viewed by others
REFERENCES
Boron Science: New Technologies and Applications, Ed. by N. S. Hosmane (CRC Press, 2011).
C. V. T. E. Hey-Hawkins, Boron-Based Compounds: Potential and Emerging Applications in Medicine (Wiley, 2018).
C. Viñas I Teixidor, Future Med. Chem. 5, 617 (2013). https://doi.org/10.4155/fmc.13.41
A. Semioshkin, A. Ilinova, I. Lobanova, et al., Tetrahedron 69, 8034 (2013). https://doi.org/10.1016/j.tet.2013.06.100
V. Bregadze, A. Semioshkin, and I. Sivaev, Appl. Radiat. Isot. 69, 1774 (2011). https://doi.org/10.1016/j.apradiso.2011.01.043
A. Semioshkin, J. Laskova, B. Wojtczak, et al., J. Organomet. Chem. 694, 1375 (2009). https://doi.org/10.1016/j.jorganchem.2008.12.024
I. M. Wyzlic, W. Tjarks, A. H. Soloway, et al., Int. J. Radiat. Oncol. 28, 1203 (1994). https://doi.org/10.1016/0360-3016(94)90495-2
G. W. Kabalka, M.-L. Yao, and Z. Wu, Org. Process Res. Dev. 10, 1059 (2006). https://doi.org/10.1021/op060052u
G. Kabalka and M.-L. Yao, Anticancer Agents Med. Chem. 6, 111 (2008).
M. J. Luderer, P. De La Puente, and A. K. Azab, Pharm. Res. 32, 2824 (2015). https://doi.org/10.1007/s11095-015-1718-y
J. D. Lee, M. Ueno, Y. Miyajima, et al., Org. Lett. 9, 323 (2007). https://doi.org/10.1021/ol062840+
A. R. Genady, Acta Chim. Slov. 59, 89 (2012).
C. N. Yeh, C. W. Chang, Y. H. Chung, et al., Eur. J. Pharm. Sci. 107, 217 (2017). https://doi.org/10.1016/j.ejps.2017.07.019
M. Y. Losytskyy, V. B. Kovalska, O. A. Varzatskii, et al., J. Lumin. 169, 51 (2016). https://doi.org/10.1016/j.jlumin.2015.08.042
F. Abi-Ghaida, S. Clément, A. Safa, et al., J. Nanomater. 2015, 1 (2015). https://doi.org/10.1155/2015/608432
I. N. Klyukin, A. P. Zhdanov, A. Y. Bykov, et al., Russ. J. Inorg. Chem. 63 (2018). https://doi.org/10.1134/S0036023618020110
J. H. Gibbs, H. Wang, N. V. S. D. K. Bhupathiraju, et al., J. Organomet. Chem. 5 (2015). https://doi.org/10.1016/j.jorganchem.2015.05.009
A. R. Genady, J. A. Ioppolo, M. M. Azaam, et al., Eur. J. Med. Chem. 93, 574 (2015). https://doi.org/10.1016/j.ejmech.2015.02.033
A. P. Zhdanov, M. V. Lisovsky, L. V. Goeva, et al., Russ. Chem. Bull. 58, 1694 (2009). https://doi.org/10.1007/s11172-009-0234-9
A. P. Zhdanov, I.N. Polyakova, G. A. Razgonyaeva, et al., Russ. J. Inorg. Chem. 56, 847 (2011). https://doi.org/10.1134/S003602361106026X
A. L. Mindich, N. A. Bokach, F. M. Dolgushin, et al., Organometallics 31 (2012). https://doi.org/10.1021/om200993f
A.L. Mindich, N.A. Bokach, M.L. Kuznetsov, et al., Organometallics 32 (2013). https://doi.org/10.1021/om400892x
A. P. Zhdanov, I. N. Klyukin, A. Y. Bykov, et al., Polyhedron 123, 176 (2017). https://doi.org/10.1016/j.poly.2016.11.035
E. A. Daines, D. S. Bolotin, N. A. Bokach, et al., Inorganica Chim. Acta 471 (2018). https://doi.org/10.1016/j.ica.2017.11.054
V. K. Burianova, D. S. Bolotin, A. S. Mikherdov, et al., New J. Chem. 42 (2018). https://doi.org/10.1039/c8nj01018h
Funding
This work has been supported by the Russian Foundation for Basic Research (project no. 19-03-00218 a) and Presidential Grant Program МD-265.2019.3.
Author information
Authors and Affiliations
Corresponding author
Supplementary material
Rights and permissions
About this article
Cite this article
Nelyubin, A.V., Klyukin, I.N., Zhdanov, A.P. et al. Synthesis of 1-Naphtylnitrilium closo-Decaborate and Amino Acid Conjugates and Their Photophysical Properties. Russ. J. Inorg. Chem. 64, 1750–1752 (2019). https://doi.org/10.1134/S0036023619140043
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619140043