Abstract
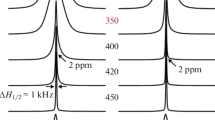
The ion mobility, phase transitions (PTs), and conductivity in (NH4)6KZr4F23 (I) and (NH4)6KHf4F23 (II) have been studied by 1H and 19F NMR, DSC, and impedance spectroscopy. Types of ion motion in the fluorides and ammonium sublattices have been determined in the temperature range 150–480 K, and their activation energies have been evaluated. Compounds I and II undergo phase transitions in the temperature ranges 400–440 and 419–440 K, respectively, to form high-temperature modifications. The high ionic conductivity in (NH4)6KZr4F23 and (NH4)6KHf4F23 (>10−3 S/cm at 473 K) makes it possible to classify these compounds with superionic conductors.
Similar content being viewed by others
References
A. V. Gerasimenko, V. Ya. Kavun, V. I. Sergienko, and T. F. Antokhina, Russ. J. Coord. Chem, 25, 562 (1999).
V. Ya. Kavun, V. I. Sergienko, N. F. Uvarov, and T. F. Antokhina, J. Struct. Chem. 43, 429 (2002).
T. F. Antokhina, L. N. Ignat’eva, N. N. Savchenko, and T. A. Kaidalova, Russ. J. Inorg. Chem. 57, 1535 (2012).
T. F. Antokhina, N. N. Savchenko, L. N. Ignat’eva, and T. A. Kaidalova, Russ. J. Coord. Chem. 39, 312 (2013).
S. P. Gabuda, Yu. V. Gagarinskii, and S. A. Polishchuk, Nuclear Magnetic Resonance in Inorganic Fluorides (Atomizdat, Moscow, 1978) [in Russian].
A. G. Lundin and E. I. Fedin, NMR Spectroscopy (Nauka, Moscow, 1986) [in Russian].
V. M. Buznik, Nuclear Spectroscopy of Inorganic Fluorides (Dal’nauka, Vladivostok, 1997) [in Russian].
T. F. Antokhina, L. N. Ignat’eva, N. N. Savchenko, and T. A. Kaidalova, Russ. J. Inorg. Chem. 49, 1314 (2004).
V. Ya. Kavun and V. I. Sergienko, Diffusion Mobility and Ion Transport in Crystalline and Amorphous Fluorides of Group IV Elements and Antimony(III) (Dal’nauka, Vladivostok, 2004) [in Russian].
A. Watton, E. C. Reynhardt, and H. E. Petch, J. Chem. Phys. 65, 4370 (1976).
V. Ya. Kavun, V. I. Sergienko, B. N. Chernyshov, et al., Zh. Neorg. Khim. 36, 1004 (1991).
V. Ya. Kavun, S. P. Gabuda, S. G. Kozlova, et al., J. Fluorine Chem. 132, 698 (2011).
V. Ya. Kavun, N. F. Uvarov, L. A. Zemnukhova, and O. V. Brovkina, Russ. J. Inorg. Chem. 49, 925 (2004).
V. Ya. Kavun, A. V. Gerasimenko, V. I. Sergienko, et al., Russ, J. Appl. Chem. 73, 1025 (2000).
V. Ya. Kavun, A. B. Slobodyuk, E. I. Voit, et al., Zh. Strukt. Khim. 51, 896 (2010).
R. E. Youngman and S. Sen, Solid State NMR 27, 77 (2005).
J. Senegas, J. M. Bobe, and J. M. Réau, Solid State Commun. 89, 983 (1994).
V. Ya. Kavun, A. V. Gerasimenko, V. I. Sergienko, et al., Russ. J. Coord. Chem. 28, 630 (2002).
S. P. Gabuda, R. L. Davidovich, S. G. Kozlova, and N. K. Moroz, Zh. Strukt. Khim. 37, 388 (1996).
V. M. Buznik, Yu. N. Moskvich, V. V. Sokolovich, et al., Zh. Strukt. Khim. 20, 622 (1979).
V. Ya. Kavun, S. P. Gabuda, S. G. Kozlova, and R. L. Davidovich, Zh. Strukt. Khim. 40, 664 (1999).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.Ya. Kavun, T.F. Antokhina, N.N. Savchenko, A.B. Podgorbunsky, T.A. Kaidalova, 2015, published in Zhurnal Neorganicheskoi Khimii, 2015, Vol. 60, No. 5, pp. 681–690.
Rights and permissions
About this article
Cite this article
Kavun, V.Y., Antokhina, T.F., Savchenko, N.N. et al. Internal mobility, phase transitions, and ionic conductivity in (NH4)6KZr4F23 and (NH4)6KHf4F23 . Russ. J. Inorg. Chem. 60, 610–618 (2015). https://doi.org/10.1134/S0036023615050095
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023615050095