Abstract
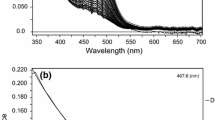
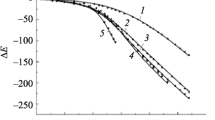
The formation constants of bismuth(III) methylthiourea complexes in aqueous solution were determined at 298 K and an acidity of 2 M HClO4 using the “ligand-oxidized ligand species” potentiometric method and the methylthiourea (mtu)-symmetric dimethylformamidine disulfide (mFDS) redox pair. The formation function was obtained, and the conditional (β * n ) and true (β n ) formation constants of Bi(mtu) 3+ n (1 ≤ n ≤ 8) were calculated. The value of β1 and the formation of complexes with coordination numbers higher than six was confirmed by spectrophotometry.
Similar content being viewed by others
References
V. Stavila, K. H. Whitmire, and I. Rusakova, Chem. Mater. 21, 5456 (2009).
R. Noel, X. Song, R. Jiang, et al., Org. Chem. 74, 7595 (2009).
K. R. Koch, Coord. Chem. Rev. 216—217, 473 (2001).
N. N. Golovnev, M. B. Egizaryan, V. A. Fedorov, et al., Russ. J. Inorg. Chem. 41, 99 (1996).
V. M. Shul’man and T. V. Kramareva, Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. 7, 55 (1961).
F. Kurzer and P. M. Sanderson, J. Chem. Soc., 4465 (1957).
V. I. Belevantsev and B. I. Peshchevitskii, Investigation of Complex Equilibria in Solutions (Nauka, Novosibirsk, 1978) [in Russian].
R. M. Smith, Critical Stability Constants. Second Supplement (Plenum, New York, 1989).
F. Hartley, C. Burgess, and R. Alcoc, Solution Equilibria (Ellis Horwood, Chichester (UK), 1980).
Cambridge Structural Database. Version 5.30 (Univ. of Cambridge, Cambridge, 2009).
J. Naslund, I. Persson, and M. Sandstrom, Inorg. Chem. 39, 4012 (2000).
Author information
Authors and Affiliations
Additional information
Original Russian Text © N.N. Golovnev, A.I. Petrov, A.O. Lykhin, A.A. Leshok, 2012, published in Zhurnal Neorganicheskoi Khimii, 2012, Vol. 57, No. 4, pp. 661–665.
Rights and permissions
About this article
Cite this article
Golovnev, N.N., Petrov, A.I., Lykhin, A.O. et al. Formation of bismuth(III) N-methylthiourea complexes in aqueous solutions. Russ. J. Inorg. Chem. 57, 596–600 (2012). https://doi.org/10.1134/S0036023612040092
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023612040092