Abstract
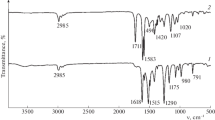
The aminated silica gels SiO2/SOCl2/NH3 (I), SiO2/SiCl4/NH3 (II), SiO2/BCl3/NH3 (III), and SiO2/γ-aminopropyltriethoxysilane (SiO2/APTES, IV) have been synthesized. According to DRIFT spectroscopy and chemical analysis data, the surface amino groups of I–III are “free,” while those of IV interact with the surface OH groups of the silica gel and with one another. The strength of basic sites has been measured on the proton affinity (PA) scale as the shift of the ν(CD) band of adsorbed deuterochloroform. The basicity of an aminated silica gel depends on its chemical composition. Silica gel IV (PA = 938 kJ/mol) is a stronger base than I–III (PA = 829 kJ/mol). As the basicity of the NH2 group decreases, the N-H stretching band shifts to higher frequencies.
Similar content being viewed by others
References
I. E. Neimark and V. M. Chertov, Dokl. Akad. Nauk SSSR 138, 877 (1961).
H. Juvaste, E. I. Liskola, and T. T. Pakkanen, J. Mol. Catal. A: Chem. 150, 1 (1999).
X. Zhang, W. Zhang, J. Li, et al., Catal. Commun. 8, 437 (2007).
Yu. V. Kholin, V. N. Zaitsev, S. A. Mernyi, et al., Ukr. Khim. Zh. 59(9), 910 (1993).
E. A. Paukshtis, N. S. Kotsarenko, and L. G. Karakchiev, React. Kinet. Catal. Lett. 12(3), 315 (1979).
Yu. V. Kholin, V. N. Zaitsev, S. A. Mernyi, et al., Ukr. Khim. Zh. 59(9), 910 (1993).
E. A. Paukshtis, Infrared Spectroscopy in Heterogeneous Acid-Base Catalysis (Nauka, Novosibirsk, 1992) [in Russian].
A. A. Chuiko, V. A. Tertykh, G. E. Plavnik, and I. E. Neimark, Kolloidn. Zh. 27(6), 903 (1965).
V. A. Tertykh, E. A. Chuiko, A. A. Chuiko, and I. E. Neimark, Kolloidn. Zh. 28(2), 278 (1966).
I. E. Neimark, V. M. Chertov, R. Yu. Sheinfain, and N. S. Kruglikova, Dokl. Akad. Nauk SSSR 132, 1356 (1960).
A. V. Kiselev, V. I. Lygin, and I. N. Solomonova, Zh. Fiz. Khim. 44(5), 1249 (1970).
K. Possemiers, P. van der Voort, and E. F. Vansant, J. Chem. Soc., Faraday Trans. 92(4), 679 (1996).
M. J. D. Low, N. Ramasubramanian, and V. V. Subba Rao, J. Phys. Chem. 71(6), 1726 (1967).
M. Folman, Trans. Faraday Soc., No. 57, 2000 (1961).
M. P. McDaniel, J. Phys. Chem. 85, 532 (1981).
L. M. Kustov, Top. Catal. 4, 131 (1997).
K. Nakanishi, Infrared Absorption Spectroscopy (Holden-Day, San Francisco, 1962; Mir, Moscow, 1965).
A. V. Kiselev and V. I. Lygin, Infrared Spectra of Surface Compounds (Nauka, Moscow, 1977) [in Russian].
L. T. Zhuravlev, Colloids Surf. A: Physicochem. Eng. Asp. 173, 1 (2000).
J. B. Peri, J. Phys. Chem. 70(9), 2937 (1966).
M. J. D. Low, A. G. Severdia, and J. Chan, J. Catal. 71, 144 (1981).
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.N. Panchenko, V.A. Pozimenko, E.A. Paukshtis, V.A. Zakharov, 2009, published in Zhurnal Neorganicheskoi Khimii, 2009, Vol. 54, No. 11, pp. 1877–1882.
Rights and permissions
About this article
Cite this article
Panchenko, V.N., Pozimenko, V.A., Paukshtis, E.A. et al. IR spectroscopic study of the basicity of aminated silica gels. Russ. J. Inorg. Chem. 54, 1798–1803 (2009). https://doi.org/10.1134/S0036023609110163
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023609110163