Abstract
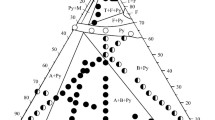
Phase relations in the Zn2V2O7-Cu2V2O7 system were studied by high-temperature X-ray diffraction and differential thermal analysis. The major phase constituents of the system are solid solutions based on Zn2V2O7 and Cu2V2O7 polymorphs and their coexistence regions. The generation of α-Zn2 − 2x Cu2x V2O7 solid solution, where 0 ≤ x ≤ 0.30, leaves almost unchanged the stabilization temperature of the high-temperature zinc pyrovanadate phase. The α-Cu2 − 2x Zn2x V2O7 homogeneity range is 5 mol % Zn2V2O7. In the range 0.050 ≤ x ≤ 0.09 from 20 to ∼ 620°C, there is the two-phase field of α-Cu2V2O7 and β-Cu2V2O7 base solid solutions. At still higher temperatures, β-Zn2 − 2x Cu2x V2O7 and α-Cu2 − 2x Zn2x V2O7 coexist in the mixed-phase region. β-Zn2 − 2x Cu2x V2O7 solid solution, where 0 ≤ x ≤ 0.30, exists above 610 ± 5°C. The extent of the β′-Cu2V2O7-base solid solution is 9 to 65 mol % Zn2V2O7 at 615 ± 5°C, expanding to 0 mol % Zn2V2O7 with rising temperature.
Similar content being viewed by others
References
M. Kurzawa, I. Rychlowska-Himmel, M. Bosacka A. Blonska-Tabero // J. Thermal Analysis and Calorimetry. 64, 1113 (2001).
N. S. Rao and O. G. Palanna, Bull. Mater. Sci. 16(1), 37 (1993).
Y. Y. Ivanova, Y. B. Dimitriev, M. R. Marinov, and D. Stavrakieva, C. R. Acad. Sci. Bulg. 27(4), 491 (1974).
G. M. Clark and R. Garlick, J. Inorg. Nucl. Chem. 40, 1347 (1978).
P. Fleury, C. R. Acad. Sci. C 263(11), 1375 (1966).
S. A. Petrova, R. G. Zakharov, M. V. Rotermel’, et al., Dokl. Akad. Nauk 400(6), 770 (2005).
R. Copal and C. Calvo, Can. J. Chem. 51, 1004 (1973).
D. Mercurio-Lavaud and B. Frit, C. R. Acad. Sci. C 277, 1101 (1873).
C. Calvo and R. Faggiani, Acta Crystallogr., Sect. B 24, 603 (1975).
T. I. Krasnenko, V. G. Zubkov, A. P. Tyutyunnik, et al., Kristallografiya 48(1), 35 (2003) [Crystallogr. Rep. 48 (1), 35 (2003)].
A. G. Nord and T. Stefanidis, Mater. Res. Bull. 20(7), 845 (1985).
J. Pommer, V. Kataev, K.-Y. Choi, et al., Phys. Rev. B: Condens. Matter 67, 214410 (2003).
A. I. Snegirev, A. A. Fotiev, V. D. Zhuravlev, and G. D. Milova, Ogneupory, No. 6, 49 (1987).
M. Schindler and F. C. Hawthorte, J. Solid State Chem. 146, 271 (1999).
M. Eguchi, M. Yamaguchi, T. Miura, and T. Kishi, Denki Kagaku 61(12), 1347 (1993).
L. A. Ponomarenko, A. N. Vasil’ev, E. V. Antipov, and Yu. A. Velikodny, Physica B 284–288, 1459 (2000).
D. de Waal and C. Hutter, Mat. Res. Bull. 29(8), 843 (1994).
A. A. Fotiev, V. K. Trunov, and V. D. Zhuravlev, Vanadates of Divalent Metals (Nauka, Moscow, 1985) [in Russian].
J. Laugier and B. Bochu, LMGP. Suite of Programs for the Interpretation of X-ray Experiments (ENSP/Laboratoire des Materiaux et du Genie Physique, BP 46. 38042 Saint Martin d’Heres, France; http://www.inpg.fr/LMGP and http://www.ccp14.ac.uk/tutorial/lmgp)
T. I. Krasnenko, B. V. Slobodin, and V. A. Zhilyaev, Zh. Neorg. Khim. 28(7), 1820 (1983).
STOE WinXPOW Version 2.06 (STOE & Cie Gmbh, Darmstadt, 2003).
Author information
Authors and Affiliations
Additional information
Original Russian Text ¢ T.I. Krasnenko, M.V. Rotermel’, S.A. Petrova, R.G. Zakharov, O.V. Sivtsova, A.N. Chvanova, 2008, published in Zhurnal Neorganicheskoi Khimii, 2008, Vol. 53, No. 10, pp. 1755–1762.
Rights and permissions
About this article
Cite this article
Krasnenko, T.I., Rotermel’, M.V., Petrova, S.A. et al. Phase relations in the Zn2V2O7-Cu2V2O7 system from room temperature to melting. Russ. J. Inorg. Chem. 53, 1641–1647 (2008). https://doi.org/10.1134/S0036023608100203
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023608100203