Abstract
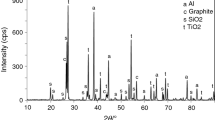
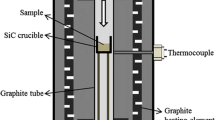
The kinetics and mechanism of the solid-phase reaction between (NH4)2SiF6 and silica at molar ratios of (0.5–5): 1 were studied. SiO2 is bound with (NH4)2SiF6 to form volatile NH4SiOF3, which abruptly enhances the ammonium hexafluorosilicate evaporation. The activation energy and rate constants of evaporation were calculated for an (NH4)2SiF6 + SiO2 (2: 1) mixture in the range 220–300°C. The reaction with crystalline SiO2 has a higher yield than with an amorphous “white soot.” The role of the SiO2 surface in the formation of the volatile products is discussed. The phase and chemical composition of the sublimates was studied.
Similar content being viewed by others
References
E. I. Mel’nichenko, G. F. Krysenko, and D. G. Epov, Zh. Neorg. Khim. 49 (12), 1943 (2004) [Russ. J. Inorg. Chem. 49 (12), 1803 (2004)].
E. I. Mel’nichenko, G. F. Krysenko, and D. G. Epov, Zh. Neorg. Khim. 50 (2), 192 (2005) [Russ. J. Inorg. Chem. 50 (2), 153 (2005)].
S. Z. Isyangulov, S. B. Savel’ev, A. A. Pupyshev, et al., RF Patent No. 2233694, Byull. Izobret., No. 22 (2004).
E. G. Rakov, Itogi Nauki Tekh., Ser.: Neorg. Khim. 15 (1988)].
R. Iler, Chemistry of Silica (Wiley, New York, 1979; Mir, Moscow, 1982).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mel’nichenko, E.I., Krysenko, G.F. & Mel’nichenko, M.N. (NH4)2SiF6 evaporation in the presence of SiO2 . Russ. J. Inorg. Chem. 51, 27–31 (2006). https://doi.org/10.1134/S0036023606010050
Received:
Issue Date:
DOI: https://doi.org/10.1134/S0036023606010050