Abstract
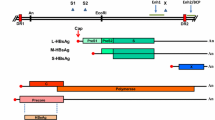
Hepatitis C virus (HCV) is a widespread dangerous human pathogen. Up to 80% of HCV-infected individuals develop chronic infection, which is often accompanied by liver inflammation and fibrosis and, at terminal stages, liver cirrhosis and cancer. Treatment of patients with end-stage liver disease is often ineffective, and even patients with suppressed HCV replication have higher risk of death as compared with noninfected subjects. Therefore, investigating the mechanisms that underlie HCV pathogenesis and developing treatments for virus-associated liver dysfunction remain an important goal. The effect of individual HCV proteins on the production of proinflammatory and profibrotic cytokines in hepatocellular carcinoma Huh7.5 cells was analyzed in a systematic manner. Cells were transfected with plasmids encoding HCV proteins. Cytokine production and secretion was accessed by immunocytochemistry and ELISA of the culture medium, and transcription of the cytokine genes was assessed using reverse transcription and PCR. HCV proteins proved to differ in effect on cytokine production. Downregulation of interleukin 6 (IL-6) production was observed in cells expressing the HCV core, NS3, and NS5A proteins. Production of transforming growth factor β1 (TGF-β1) was lower in cells expressing the core proteins, NS3, or E1/E2 glycoproteins. A pronounced increase in production and secretion of tumor necrosis factor α (TNF-α) was observed in response to expression of the HCV E1/E2 glycoproteins. A higher biosynthesis, but a lower level in the cell culture medium, was detected for interleukin 1β (IL-1β) in cells harboring NS4 and IL-6 in cells expressing NS5В. The finding was possibly explained by protein-specific retention and consequent accumulation of the respective cytokines in the cell.
Similar content being viewed by others
Abbreviations
- IFN:
-
interferon
- IL:
-
interleukin
- NS:
-
nonstructural protein
- TGF:
-
transforming growth factor
- TNF:
-
tumor necrosis factor
- HCV:
-
hepatitis C virus
- ELISA:
-
enzyme-linked immunosorbent assay
- mAB:
-
monoclonal antibody
- DAA:
-
direct-acting antiviral agent
References
Li H.C., Lo S.Y. 2015. Hepatitis C virus: Virology, diagnosis and treatment. World J. Hepatol. 7, 1377–1389.
Verstrepen B.E., Boonstra A., Koopman G. 2015. Immune mechanisms of vaccine induced protection against chronic hepatitis C virus infection in chimpanzees. World J. Hepatol. 7, 53–69.
Szabo G., Petrasek J. 2015. Inflammasome activation and function in liver disease. Nat. Rev. Gastroenterol. Hepatol. 12, 387–400.
Gondeau C., Briolotti P., Razafy F., Duret C., Rubbo P.A., Helle F., Reme T., Ripault M.P., Ducos J., Fabre J.M., Ramos J., Pecheur E.I., Larrey D., Maurel P., Daujat- Chavanieu M. 2014. In vitro infection of primary human hepatocytes by HCV-positive sera: Insights on a highly relevant model. Gut. 63, 1490–1500.
Zampino R., Marrone A., Restivo L., Guerrera B., Sellitto A., Rinaldi L., Romano C., Adinolfi L.E. 2013. Chronic HCV infection and inflammation: Clinical impact on hepatic and extra-hepatic manifestations. World J. Hepatol. 5, 528–540.
Negash A.A., Ramos H.J., Crochet N., Lau D.T., Doehle B., Papic N., Delker D.A., Jo J., Bertoletti A., Hagedorn C.H., Gale M., 2013. IL-1ß production through the NLRP3 inflammasome by hepatic macrophages links hepatitis C virus infection with liver inflammation and disease. PLoS Pathog. 9, e1003330.
Brenner C., Galluzzi L., Kepp O., Kroemer G. 2013. Decoding cell death signals in liver inflammation. J. Hepatol. 59, 583–594.
Preßser L.D., Haskett A., Waris G. 2011. Hepatitis C virus-induced furin and thrombospondin-1 activate TGF-ß1: Role of TGF1 in HCV replication. Virology. 412, 284–296.
Bonilla N., Barget N., Andrieu M., Roulot D., Letoumelin P., Grando V., Trinchet J., Ganne-Carrie N., Beaugrand M., Deny P., Choppin J., Guillet J., Ziol M. 2006. Interferon gamma-secreting HCV-specific CD8+ T cells in the liver of patients with chronic C hepatitis: Relation to liver fibrosis-ANRS HCEP07 study. J. Viral Hepat. 13, 474–481.
Basu A., Meyer K., Lai K.K., Saito K., Di Bisceglie A.M., Grosso L.E., Ray R.B., Ray R. 2006. Microarray analyses and molecular profiling of Stat3 signaling pathway induced by hepatitis C virus core protein in human hepatocytes. Virology. 349, 347–358.
Yu G.Y., He G., Li C.Y., Tang M., Grivennikov S., Tsai W.T., Wu M.S., Hsu C.W., Tsai Y., Wang L.H., Karin M. 2012. Hepatic expression of HCV RNAdependent RNA polymerase triggers innate immune signaling and cytokine production. Mol. Cell. 48, 313–321.
Collins A.S., Ahmed S., Napoletano S., Schroeder M., Johnston J.A., Hegarty J.E., O’ Farrelly C., Stevenson N.J. 2014. Hepatitis C virus (HCV)-induced suppressor of cytokine signaling (SOCS) 3 regulates proinflammatory TNF-a responses. J. Leukoc. Biol. 96, 255–263.
Lindenbach B.D., Evans M.J., Syder A.J., Wölk B., Tellinghuisen T.L., Liu C.C., Maruyama T., Hynes R.O., Burton D.R., McKeating J.A., Rice C.M. 2005. Complete replication of hepatitis C virus in cell culture. Science. 309, 623–626.
Wakita T., Pietschmann T., Kato T., Date T., Miyamoto M., Zhao Z., Murthy K., Habermann A., Kräusslich H.G., Mizokami M., Bartenschlager R., Liang T.J. 2005. Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat. Med. 11, 791–796.
Zhong J., Gastaminza P., Cheng G., Kapadia S., Kato T., Burton D.R., Wieland S.F., Uprichard S.L., Wakita T., Chisari F.V. 2005. Robust hepatitis C virus infection in vitro. Proc. Natl. Acad. Sci. U. S. A. 102, 9294–9299.
Masalova O.V., Lesnova E.I., Ivanov A.V., Pichugin A.V., Permyakova K.Yu., Smirnova O.A., Tynitskaia V.L., Ulanova T.I., Burkov A.N., Kochetkov S.N., Ataullakhanov R.I., Kushch A.A. 2013. Comparative analysis of the immune response to DNA constructions encoding hepatitis C virus nonstructural proteins. Vopr. Virusol. 58, 21–28.
Permyakova K.Yu., Lesnova E.I., Masalova O.V., Ivanov A.V., Ataullakhanov R.I., Kushch A.A. 2015. Immunogenic properties of DNA constructs containing structural and nonstructural regions of hepatitis C virus. Immunologiya. 36, 162–167.
Ivanov A.V., Smirnova O.A., Ivanova O.N., Masalova O.V., Kochetkov S.N., Isaguliants M.G. 2011. Hepatitis C virus proteins activate NRF2/ARE pathway by distinct ROS-dependent and independent mechanisms in HUH7 cells. PLoS ONE. 6, e24957.
Masalova O.V., Lesnova E.I., Grabovetskii V.V., Smirnova O.A., Ulanova T.I., Burkov A.N., Ivanov A.V., Zaberezhnyi A.D., Ataullakhanov R.I., Kushch A.A. 2010. DNA immunization with a plasmid carrying the gene of hepatitis C virus protein 5A (NS5A) induces an effective cellular immune response. Mol. Biol. (Moscow). 44 (2), 245–253.
Rechkina E.A., Denisova G.F., Masalova O.V., Lideman L.F., Denisov D.A., Lesnova E.I., Ataullakhanov R.I., Gurianova S.V., Kushch A.A. 2006. Mapping of antigenic determinants of hepatitis C virus proteins using phage display. Mol. Biol. (Moscow). 40 (2), 312–323.
Masalova O.V., Abdulmedzhidova A.G., Shkurko T.V., Kelli E.I., Atanadze S.N., Zavalishina L.E., Frank G.A., Kuzina O.V., L’ vov D.K., Kushch A.A. 2003. Analysis of hepatitis C virus proteins in the liver of patients with chronic hepatitis C. Vopr. Virusol. 48, 9–14.
Vishnevskaya T.V., Masalova O.V., Al’ khovskii S.V., Pichugin A.V., Shkurko T.V., Kelli E.I., Ataullakhanov R.I., Blokhina N.P., Kushch A.A. 2008. Detection of hepatitis C virus replication markers in peripheral blood mononuclear cells of patients with chronic hepatitis C. Med. Immunol. 10, 397–404.
Hanafusa H., Morikawa Y., Uehara T., Kaneto M., Ono A., Yamada H., Ohno Y., Urushidani T. 2014. Comparative gene and protein expression analyses of a panel of cytokines in acute and chronic drug-induced liver injury in rats. Toxicology. 324, 43–54.
Hassan M., Selimovic D., Ghozlan H., Abdel-Kader O. 2007. Induction of high-molecular-weight (HMW) tumor necrosis factor (TNF) alpha by hepatitis C virus (HCV) non-structural protein 3 (NS3) in liver cells is AP-1 and NF-kappaB-dependent activation. Cell. Signal. 19, 301–311.
Lin Y.T., Wu Y.H., Tseng C.K., Lin C.K., Chen W.C., Hsu Y.C., Lee J.C. 2013. Green tea phenolic epicatechins inhibit hepatitis C virus replication via cycloxygenase- 2 and attenuate virus-induced inflammation. PLoS ONE. 8, e54466.
Benzoubir N., Lejamtel C., Battaglia S., Testoni B., Benaßsi B., Gondeau C., Perrin-Cocon L., Desterke C., Thiers V., Samuel D., Levrero M., Bréchot C., Bourgeade M.F. 2013. HCV core-mediated activation of latent TGF via thrombospondin drives the crosstalk between hepatocytes and stromal environment. J. Hepatol. 59, 1160–1168.
Ivanov A.V., Smirnova O.A., Petrushanko I.Y., Ivanova O.N., Karpenko I.L., Alekseeva E., Sominskaya I., Makarov A.A., Bartosch B., Kochetkov S.N., Isaguliants M.G. 2015. HCV core protein uses multiple mechanisms to induce oxidative stress in human hepatoma HUH7 cells. Viruses. 7, 2745–2770.
Lee J., Tian Y., Chan S.T., Kim J.Y., Cho C., Ou J.H. 2015. TNF-a induced by hepatitis C virus via TLR7 and TLR8 in hepatocytes supports interferon signaling via an autocrine mechanism. PLoS Pathog. 11, e1004937.
Nishitsuji H., Funami K., Shimizu Y., Ujino S., Sugiyama K., Seya T., Takaku H., Shimotohno K. 2013. Hepatitis C virus infection induces inflammatory cytokines and chemokines mediated by the cross talk between hepatocytes and stellate cells. J. Virol. 87, 8169–8178.
Guerriero E., Capone F., Rusolo F., Colonna G., Castello G., Costantini S. 2013. Dissimilar cytokine patterns in different human liver and colon cancer cell lines. Cytokine. 64, 584–589.
Alborzi A.M., Bamdad T., Davoodian P., Hashempoor T., Nejatizadeh A.A., Moayedi J. 2015. Insights into the role of HCV Plus/Minus strand RNA, IFN and IL-29 in relapse outcome in patients infected with HCV. Asian Pac. J. Allergy Immunol. 33, 173–181
Nakamoto S., Kanda T., Shirasawa H., Yokosuka O. 2015. Antiviral therapies for chronic hepatitis C virus infection with cirrhosis. World J. Hepatol. 7, 1133–1141.
Attar B.M., van Thiel D. 2015. A new twist to a chronic HCV infection: Occult hepatitis C. Gastroenterol. Res. Pract. 2015, 579147.
Selimovic D., El-Khattouti A., Ghozlan H., Haikel Y., Abdelkader O., Hassan M. 2012. Hepatitis C virusrelated hepatocellular carcinoma: An insight into molecular mechanisms and therapeutic strategies. World J. Hepatol. 4, 342–355.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.V. Masalova, E.I. Lesnova, K.Yu. Permyakova, E.I. Samokhvalov, A.V. Ivanov, S.N. Kochetkov, A.A. Kushch, 2016, published in Molekulyarnaya Biologiya, 2016, Vol. 50, No. 3, pp. 486–495.
Rights and permissions
About this article
Cite this article
Masalova, O.V., Lesnova, E.I., Permyakova, K.Y. et al. Effect of Hepatitis C virus proteins on the production of proinflammatory and profibrotic cytokines in Huh7.5 human hepatoma cells. Mol Biol 50, 422–430 (2016). https://doi.org/10.1134/S0026893316020163
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893316020163