Abstract
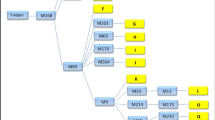
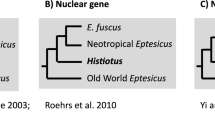
Four types of nuclear DNA markers identified by the taxonprint, RAPD, and IMP (Inter-MIR-PCR) methods, and the nucleotide sequences of satellite DNA monomers have been used to analyze the molecular genetic similarity between some populations, subspecies, and species of lizards combined into the group Lacerta s. str., as well as representatives of some other genera. The notions on the systematics and phylogeny of this group based on morphological and zoogeographic criteria have been compared to the conclusions based on molecular genetic data. The genus and species subdivisions of populations based on nuclear molecular markers and morphological characters generally agree with each other, the degree of genetic differences being correlated with the taxonomy suggested by zoomorphologists. The degree of differences between the subspecies of one of the species studied, Lacerta agilis, varies depending on the molecular markers used: according to the results of RAPD analysis, all subspecies substantially differ from one another, the variation within populations being small; with respect to other markers, the differences are smaller and not equivalent. The existence of the so-called eastern and western clades of this species earlier assumed by other researchers on the basis of mtDNA and morphological data has been confirmed. There are no distinct gradations exceeding individual variation in 14 populations of L. agilis exigua (the eastern clade) with respect to IMP markers, although these populations inhabit a vast area from the Ural Mountains to the Kabardino-Balkar Republic (the Caucasus). These data suggest that the subspecies has been rapidly spreading northwards since the Pleistocene glaciation (about 15,000 years ago).
Similar content being viewed by others
References
Stewart C.-B. 1993. The powers and pittfalls of parsimony. Nature. 361, 603–607.
Antonov A.S. 2000. Osnovy genosistematiki rastenii (Findamentals of Plant Genosystematics). Moscow: MAIK Nauka/Interperiodica.
Baker R.H., Gatesy J. 2002. Is morphology still relevant? In: Molecular Systematics and Evolution: Theory and Practice. Eds. DeSalle R., Giribe G., Wheeler W. Basel: Birkhauser, pp. 163–174.
Molecular Systematics. Eds. Hillis D.M., Moritz C., Mable B.K., Sunderland, MA: Sinauer, 1996.
Tautz D., Arctander P., Minelli A., Thomas R.H., Voger A.P. 2003. A plea for DNA taxonomy. Trends Ecol. Evol. 18, 70–74.
Lee M.S.Y. 1999. Molecular phylogeny becomes functional. Trends Ecol. Evol. 14, 177–178.
Nichols R.A. 2001. Gene tree and species tree are not the same. Biol. J. Linn. Soc. 68, 87–112.
Emelyanov A.F., Rasnitsyn A.P. 1991. Systematics, Phylogeny, and Cladistics. Priroda. 7, 23–37.
Elder J.F., Turner B.J. 1995. Concerted evolution of repetitive DNA sequences in eukaryotes. Quant. Rev. Biol. 70, 297–320.
Grechko V.V. 2002. Molecular markers in phylogeny and systematics. Genetika. 39, 1527–1541.
Hillis D.M. 1987. Molecular versus morphological approaches in evolution. Annu. Rev. Ecol. Syst. 18, 23–42.
Avise J.C. 2004. Molecular Markers, Natural History, and Evolution. 2nd ed. Sunderland, MA: Sinauer.
Brower A.V.Z., DeSalle R., Vogler A. 1996. Gene tree, species tree, and systematics. Annu. Rev. Ecol. Syst. 27, 423–450.
Hewitt G.M. 1999. Post-glacial re-colonization of European biota. Biol. J. Linn. Soc. 68, 87–112.
Darevsky I.S., Shcherbak N.N., Peters T., et al. 1976. Systematics and intraspecific structure. In: Prytkaya yashcheritsa (The Sand Lizard). Ed. Yablokov A.V. Moscow: Nauka, pp. 53–68.
Bischoff W. 1991. Ubersicht der Arten and interarten der Familie Lacertidae. 3. Die Gattung Lacerta. In: Die Eidechse, Mittellungblatt der AG Lacertiden in der DGHT. Hefte 3. Bonn/Bremen, 5–18.
Kalyabina-Hauf S.A., Ananyeva N.B. 2004. Phylogeny and intraspecific structure of Lanarta agilis L. 1758 (Lanartidaa, Sauria, Reptilia), a lizard species with a broad range (experience in using the mitochondrial cytochrome b gene). Tr. Zool. Inst. Ross. Akad. Nauk. 32, 1–108.
Arnold E.N. 1989. Towards a phylogeny and biogeography of the Lacertidae: Relationships within an Old-World family of lizards derived from morphology. Bull. Br. Mus (Nat. Hist.) Zool. 55, 209–257.
Arribas O.J. 1999. Phylogeny and relationships of the mountain lizards of Europe and Near East (Archaeolacerta Mertens, 1921, sensu lato) and their relationships among the Eurasian lacertid radiation. Russ. J. Herpetol. 16, 1–22.
Harris D.J. 1999. Molecular systematics and evolution of lacertid lizards. Nature Croat. 8, 161–180.
Harris D.J., Arnold E.N., Thomas R. 1998. Relationships of lacertid lizards (Reptilia: Lacertidae) estimated from mitochondrial DNA sequences and morphology. Proc. R. Soc. Lond. B. 265, 1939–1958.
Kalyabina S.A., Milto K.D., Ananyeva N.B., Legal L., Joger U., Wink H. 2001. Phylogeography and systematics of Lacerta agilis based on mitochondrial cytochrome b gene sequence: First results. Russ. J. Herpetol. 8, 149–158.
Grechko V.V., Fedorova L.V., Fedorov A.N., et al. 1997. Restriction endonuclease analysis of highly repetitive DNA as a phylogenetic tool. J. Mol. Evol. 45, 332–336.
Fedorov A.N., Fedorova L.V., Grechko V.V., et al. 1999. Variable and invariable DNA repeat characters revealed by taxonprint approach are useful for molecular systematics. J. Mol. Evol. 48, 69–76.
Ryabinina N.L., Bannikova A.A., Kosushkin S.A., et al. 2002. Estimation of the subspecies level of differentiation in Caucasian lizards of the genus Darevskia (syn. “Lacreta saxicola complex”, Lacertidae, Sauria) using genome DNA markers. Russ. J. Herpetol. 9, 185–194.
Borodulina O.R., Kramerov D.A. 2005. PCR-based approach to SINE isolation: Simple and complex SINEs. Gene. 349, 197–205.
Ryabinina N.L., Grechko V.V., Darevsky I.S. 1998. DNA polymorphism detectable by RAPD analysis in populations of Lacertidae lizards. Genetika. 34, 1661–1667.
Ciobanu D., Grechko V.V., Darevsky I.S. 2003. Molecular evolution of NLsat satellite DNA in lizards of the genus Daravskia (Sauria: Lacertidae): Correlation with species diversity. Genetika. 39, 1527–1541.
Ciobanu D.G., Grechko V.V., Darevsky I.S., Kramerov D.A. 2004. New satellite DNA in Lacerta s. str. lizards (Sauria: Lacertidae): Evolutionary pathways and phylogenetic impact. J. Exp. Zool. 302B, 505–516.
Grechko V.V., Ciobanu D.G., Darevsky I.S., Kramerov D.A. 2005. Satellite DNA of the genus Lacerta s. str. (L. agilis group), Lacertidae. Dokl. Akad. Nauk. 400, 392–395.
Kimura M. 1980. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 16, 111–120.
Van de Peer Y., De Wachter R. 1994. TREECON for Windows: A software package for construction and drawing of evolution trees for Microsoft Windows environment. Comput. Applic. Biosci. 10, 569–570.
Grechko V.V., Ryabinin D.M., Fedorov A.N., Rudykh A.I., Ryskov A.P., Semenova S.K., Darevsky I.S. 1998. Molecular genetic classification and phylogenetic relatedness of some lacertid lizard species as inferred from studies on specific distribution of restriction sites in DNA repeats (taxonprinting). Mol. Biol. 36, 172–183.
Fu J. 2000. Toward the phylogeny of the family Lacertidae: Why 4798 base pairs of mtDNA sequences cannot draw the picture. Biol. J. Linn. Soc. 71, 203–217.
Ryabinin D.M., Grechko V.V., Darevsky I.S., Ryskov A.P., Semenova S.K., 1996. Comparative study of DNA repetitive sequences by means of restriction endonucleases among populations and subspecies of some lacertid lizard species. Russ. J. Herpetol. 3, 178–185.
Ciobanu D.G., Grechko V.V., Kramerov D.A. 2003. A new subfamily of satellite DNA, NLsatIV, in Daravskia lindhilmi lizards (Lacertidae): Structure and evolution. Dokl. Akad. Nauk. 392, 1–5.
Ciobanu D.G., Rudykh I.A., Ryabinina N.L., Grechko V.V., Kramerov D.A., Darevsky I.S. 2002. Reticulate evolution of parthenogenetic rock lizard species (Lacertidae): Inheritance of tandem repeats and anonymous RAPD markers. Mol. Biol. 36, 296–306.
Gilbert W., Labuda D. 1999. CORE-SINE: Eukaryotic short interspersed retroposone elements with common sequence motifs. Proc. Natl. Acad. Sci. USA. 96, 2869–2874.
Nichols R.A., Hewitt G.M. 1994. The genetic cousequencies of long distance dispersal during colonization. Heredity. 72, 312–317.
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.V. Grechko, L.V. Fedorova, D.M. Ryabinin, N.L. Ryabinina, D.G. Ciobanu, S.A. Kosushkin, I.S. Darevsky, 2006, published in Molekulyarnaya Biologiya, 2006, Vol. 40, No. 1, pp. 61–73.
If a gene tree conflicts with an accepted tree, one should stop and ponder why. C. B. Stewart [1].
Rights and permissions
About this article
Cite this article
Grechko, V.V., Fedorova, L.V., Ryabinin, D.M. et al. The use of nuclear DNA molecular markers for studying speciation and systematics as exemplified by the “Lacerta agilis complex” (Sauria: Lacertidae). Mol Biol 40, 51–62 (2006). https://doi.org/10.1134/S0026893306010092
Received:
Issue Date:
DOI: https://doi.org/10.1134/S0026893306010092