Abstract
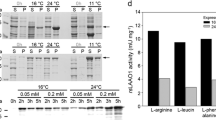
L-lysine-α-oxidase (LO) synthesized by the fungus Trichoderma cf. aureoviride Rifai VKM F-4268D under salt stress conditions was isolated and characterized. The newly developed method for the isolation and purification of the enzyme was based on its precipitation from the culture liquid by copper sulfate. The subsequent LO purification by the methods of hydrophobic (Octyl Sepharose) and ion exchange (DEAE ToyoPearl) chromatography yielded a homogeneous enzyme preparation with a high degree of purification (310-fold) and high specific activity (90 U/mg protein). The molecular mass of the enzyme determined by gel filtration and native electrophoresis was 115–116 kDa. According to the data of SDS electrophoresis, LO was a dimer with identical subunits (57–58 kDa). The optical absorption spectrum of LO corresponded to the flavoprotein spectrum with maximums at 278, 390, and 465 (a shoulder at 490) nm. LO is a stereospecific enzyme oxidizing almost exclusively L-lysine (pH optimum 7.8–8.2). Insignificant activity was observed against L-ornithine and L-arginine. LO was shown to be stable at temperatures up to 50°C.
Similar content being viewed by others
References
Berezov, T.T., Molecular and Biochemical Bases of Enzyme Therapy of Tumors, Biomed. Khim., 2005, vol. 51, no. 3, pp. 235–247.
Lukasheva, E.V. and Berezov, T.T., L-Lysine α-Oxidase: Physicochemical and Biological Properties, Biochemistry (Moscow), 2002, vol. 67, no. 10, pp. 1152–1158.
Murthy, S.N. and Janardanasarma, M.K., Identification of L-Amino Acid/L-Lysine Alpha-Amino Oxidase in Mouse Brain, Mol. Cell. Biochem., 1999, vol. 197, no. 1–2, pp. 13–23.
Sun, Y., Nonobe, E., Kobayashi, Y., Kuraishi, T., Aoki, F., Yamamoto, K., and Sakai, S., Characterization and Expression of L-Amino Acid Oxidase of Mouse Milk, J. Biol. Chem., 2002, vol. 277, no. 21, pp. 19080–19086.
Du, X.Y. and Clementson, K.J., Snake Venom L-Amino Acid Oxidases, Toxicon, 2002, vol. 40, no. 6, pp. 659–665.
Ahn, M.Y., Ryu, K.S., Lee, Y.W., and Kim, Y., Cytotoxicity and L-Amino Acid Oxidase Activity of Crude Insect Drugs, Arch. Pharm. Res., 2000, vol. 23, pp. 477–481.
Nagashima, Y., Tsukamoto, C., Kitani, Y., Ishizaki, S., Nagai, H., and Yanagimoto, T., Isolation and cDNA Cloning of an Antibacterial L-Amino Acid Oxidase from the Skin Mucus of the Great Sculpin Myoxocephalus polyacanthocephalus, Comp. Biochem. Physiol. B, 2009, no. 154, pp. 55–61.
Kitani, Y., Tsukamoto, C., Zhang, G., Nagai, H., Ishida, M., Ishizaki, S., Shimakura, K., Shiomi, K., and Nagashima, Y., Identification of an Antibacterial Protein as L-Amino Acid Oxidase in the Skin Mucus of Rockfish Sebastes schlegeli, FEBS J., 2007, vol. 274, no. 1, pp. 125–136.
Chen, W.M., Sheu, S., and Sheu, S.Y., Novel L-Amino Acid Oxidase with Algicidal Activity against Toxic Cyanobacterium Microcystis aeruginosa Synthesized by a Bacterium Aquimarina sp., Enzyme Microb. Technol., 2011, vol. 49, no. 4, pp. 372–379.
Gomez, D., Lucas-Elio, P., Sanchez-Amat, A., and Solano, F., A Novel Type of Lysine Oxidase: L-Lysine-Epsilon-Oxidase, Biochim. Biophys. Acta, 2006, vol. 1764, no. 10, pp. 1577–1585.
Böhmer, A., Müller, A., Passarge, M., Liebs, P., Honeck, H., and Müller, H., A Novel L-Glutamate Oxidase from Streptomyces endus. Purification and Properties, Eur. J. Biochem., 1989, vol. 182, no. 2, pp. 327–332.
Koyama, H., Purification and Characterization of a Novel L-Phenylalanine Oxidase (Deaminating and Decarboxylating) from Pseudomonas sp. P-501, J. Biochem., 1982, vol. 92, pp. 1235–1240.
Calderon, J., Olvera, I., Martinez, M., and Davila, G., A Neurospora crassa Mutant Altered in the Regulation of L-Amino Acid Oxidase, Microbiolology (UK), 1997, vol. 143, pp. 1969–1974.
Nuutinen, J.T. and Timonen, S., Identification of Nitrogen Mineralization Enzymes, L-Amino Acid Oxidases, from the Ectomycorrhizal Fungi Hebeloma spp. and Laccaria bicolor, Mycol. Res., 2008, vol. 112, no. 12, pp. 1453–1464.
Singh, S., Gogoi, B.K., and Bezbaruah, R.L., Optimization of Medium and Cultivation Conditions for L-Amino Acid Oxidase Production by Aspergillus fumigatus, Can. J. Microbiol., 2009, vol. 55, no. (9), pp. 1096–1102.
Nuutinen, J.T., Marttinen, E., Soliymani, R., Hildén, K., and Timonen, S., L-Amino Acid Oxidase of the Fungus Hebeloma cylindrosporum Displays Substrate Preference Towards Glutamate, Microbiology (UK), 2012, vol. 158, no. 1, pp. 272–283.
Kusakabe, H., Kodama, K., Kuninaka, A., Yoshino, H., Misono, H., and Soda, K., A New Antitumor Enzyme, L-Lysine Alpha-Oxidase from Trichoderma viride. Purification and Enzymological Properties, J. Biol. Chem., 1980, vol. 255, no. 3, pp. 976–981.
Kusakabe, H., Kodama, K., Kuninaka, A., Yoshino, H., and Soda, K., Extracellular Production L-Lysine α-Oxidase in Wheat Bran Culture of a Strain of Trichoderma viride, Agric. Biol. Chem., 1979, vol. 43, no. 12, pp. 2531–2535.
Weber, E., Tonder, K., Reinboth, C., Unverhau, K., Weide, H., and Aurich, H., L-Lysin-Oxidase from Trichoderma viride i4. Purification and Characterization, J. Bas. Microbiol., 1990, vol. 34, pp. 265–276.
Makrushin, K.V., Arinbasarova, A.Yu., Lukasheva, E.V., Medentsev, A.G., and Berezov, T.T., Search for Fungal Producers of L-Lysine-α-Oxidase, Biotekhnologiya, 2011, no. 5, pp. 44–49.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.Yu. Arinbasarova, V.V. Ashin, K.V. Makrushin, A.G. Medentsev, E.V. Lukasheva, T.T. Berezov, 2012, published in Mikrobiologiya, 2012, Vol. 81, No. 5, pp. 594–599.
Rights and permissions
About this article
Cite this article
Arinbasarova, A.Y., Ashin, V.V., Makrushin, K.V. et al. Isolation and properties of L-lysine-α-oxidase from the fungus Trichoderma cf. aureoviride RIFAI VKM F-4268D. Microbiology 81, 549–554 (2012). https://doi.org/10.1134/S0026261712050037
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261712050037