Abstract
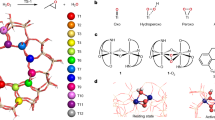
The high activity of titanium silicate (TS-1) in oxidations with hydrogen peroxide is the result of different factors, directly referable to the MFI structure. The configuration of Ti site (tetrahedral), the nature of active species (Ti-OOH) and the nature of the surface (hydrophobic) are the three factors that are immediately apparent. Hydrophobicity alone, however, cannot fully explain certain catalytic performances and trends in the oxidation of the olefins, without envisaging a major involvement of porosity. Actually, the molecular dimensions of pores (ca. 0.55 nm), normally perceived as just a drawback for the limits set to the range of potential substrates, play a relevant and positive role in TS-1 catalysis. The purpose of the paper is to illustrate and discuss these issues, with supporting evidences mostly taken from the oxidation of olefins and alkanes.
Similar content being viewed by others
References
Liquid Phase Oxidation via Heterogeneous Catalysis: Organic Synthesis and Industrial Applications, Clerici, M.G. and Kholdeeva, O.A., Eds., Hoboken, N.J.: Wiley, 2012.
Clerici, M.G., Appl. Catal., 1991, vol. 68, p. 249.
Bellussi, G., Carati, A., Clerici, M.G., Maddinelli, G., and Millini, R., J. Catal., 1992, vol. 133, p. 220.
To, J., Sokol, A.A., French, S.A., and Catlow, C.R.A., J. Phys. Chem. C, 2008, vol. 112, p. 7173.
Clerici, M.G., Metal Oxide Catalysis, Jackson S.D. and Hargreaves, J.S.J., Eds., Weinheim: Wiley-VCH, 2009, vol. 2, p. 705.
Flanigen, E.M., Bennett, J.M., Grose, R.W., Cohen, J.P., Patton, R.L., and Kirchner, R.M., Nature, 1978, vol. 271, no. 5645, p. 512.
Weitkamp, J., Ernst, S., Roland, E., and Thiele, G.F., Stud. Surf. Sci. Catal., 1997, vol. 105, p. 763.
Clerici, M.G., Bellussi, G., and Romano, U., J. Catal., 1991, vol. 129, p. 159.
Clerici, M.G. and Ingallina, P., J. Catal., 1993, vol. 140, p. 71.
Blasco, T., Camblor, M.A., Corma, A., Esteve, P., Martinez, A., Prieto, C., and Valencia, S., Chem. Commun., 1996, p. 2367.
Derouane, E.G., J. Mol. Catal. A, 1998, vol. 134, p. 29.
Derouane, E.G., Chem. Phys. Lett., 1987, vol. 142, nos. 3–4, p. 200.
Langhendries, G., De Vos, D.E., Baron, G.V., and Jacobs, P.A., J. Catal., 1999, vol. 187, p. 453.
Van der Waal, J.C., Rigutto, M.S., and van Bekkum, H., Appl. Catal., A, 1998, vol. 167, p. 331.
Corma, A., Esteve, P., and Martínez, A., J. Catal., 1996, vol. 161, p. 11.
Clerici, M.G., Top. Catal, 2001, vol. 15, p. 257.
Antonova, N.S., Carbó, J.J., Kortz, U., Kholdeeva, O.A., and Poblet, J.M., J. Am. Chem. Soc., 2010, vol. 132, p. 7488.
Yoon, C.W., Hirsekorn, K.F., Neidig, M.L., Yang, X.Z., and Tilley, T.D., ACS Catal., 2011, vol. 1, p. 1665.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Clerici, M.G. The activity of titanium silicalite-1 (TS-1): Some considerations on its origin. Kinet Catal 56, 450–455 (2015). https://doi.org/10.1134/S0023158415040059
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158415040059