Abstract
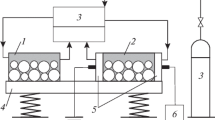
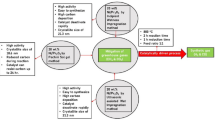
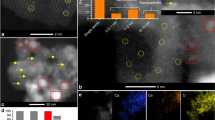
The nanopowders of Ni and Co metals and their alloy were prepared by plasma-assisted milling, and the activity of catalysts on their basis was studied in the carbon dioxide reforming of methane (CDRM) at atmospheric pressure. It was found that the catalytic activity of Ni nanopowder rapidly decreased because of the blocking of its surface by coking products. Co powder exhibited lower but stable activity, which gradually decreased as a result of coking only 300 h after the onset of reaction. A Ni-Co alloy (1: 1, by weight) is an active and selective catalyst for CDRM. Its catalytic activity appeared at 400°C; at 870°C, conversion reached 90% and remained unchanged for 500 h. The initial activity was restored by the regeneration of a catalyst based on the Ni-Co alloy with molecular hydrogen for several hours at 400°C.
Similar content being viewed by others
References
Tret’yakov, Yu.D., Nanotekhnologii (Nanotechnologies), Moscow: Fizmatlit, 2009.
Gusev, A.I., Nanomaterialy, nanostruktury, nanotekhnologii (Nanomaterials, Nanostructures, and Nanotechnologies), Moscow: Fizmatlit, 2007.
Krylov, O.V., Ross. Khim. Zh., 2000, vol. 44, no. 1, p. 19.
Ma, J., Sun, N., Zhang, X., Zhao, N., Xiao, F., Wei, W., and Sun, Y., Catal. Today, 2009, vol. 148, p. 221.
Guo, J., Lou, H., Zhao, H., Chai, D., and Zheng, X., Appl. Catal., A, 2004, vol. 273, p. 75.
Halliche, D., Bauarab, R., Cherifi, O., and Bettahar, M.M., Catal. Today, 1996, vol. 29, no. 1.
Liu, D., Ni, W., Cheo, E., Wen, Y., Lim, Y., Borgna, A., Lau, R., and Yang, Y., Catal. Today, 2010, vol. 154, nos. 3–4, p. 229.
Rujing Shang, Xiaoning Guo, Shifang Mu, Yingyong Wang, Guoqiang Jin, Hendrik Kosslick, Axel Schulz, and Xiang-Yun Guo, Int. J. Hydrogen Energy, 2011, vol. 36, no. 8, p. 4900.
Fan, M.S., Abdullah, A.Z., and Bhatia, S., Int. J. Hydrogen Energy, 2011, vol. 36, no. 8, p. 4875.
Claridge, J.B., York, A.P.E., Brungs, A.J., MarquezAlvarez, C., Sloan, J., Tsang, Sh.C., and Green, M.L.H., J. Catal., 1998, vol. 180, p. 85.
Sehested, J., Jacobsen, C.J.H., Rokni, S., and Rostrup-Nielsen, J.R., J. Catal., 2001, vol. 201, p. 206.
Grigoryan, R.R., Aloyan, S.G., Vartikyan, L.A., Saakyan, M.S., and Arutyunyan, V.G., Tezisy dokl. 2-oi mezhd. konf. po khimii i khimicheskoi tekhnologii (Proc. 2nd Int. Conf. on Chemistry and Chemical Engineering), Yerevan, 2010, p. 295.
Grigoryan, R.R., Vartikyan, L.A., Mnatsakanyan, R.A., and Zurnachyan, A.R., Chem. J. Armenia, 2010, vol. 63, p. 535.
Sheldon, R.A., Chemicals from Synthesis Gas, Dordrecht: Reidel, 1983.
Arutyunov, V.S., Ross. Khim. Zh., 2001, vol. 45, no. 1, p. 55.
Calka, A. and Wexler, D., Mater. Sci. Forum, 2002, vols. 368–388, p. 125.
Er-rbib, H., Bouallou, C., and Werkoff, F., Chem. Eng. Trans., 2012, vol. 29, p. 163.
Molchanov, V.V. and Buyanov, R.A., Russ. Che. Rev., 2000, vol. 69, no. 5, p. 435.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © R.R. Grigoryan, L A. Vartikyan, L.A. Tavadyan, S.G. Aloyan, V.R. Arutyunyan, 2014, published in Kinetika i Kataliz, 2014, Vol. 55, No. 2, pp. 234–238.
Rights and permissions
About this article
Cite this article
Grigoryan, R.R., Vartikyan, L.A., Tavadyan, L.A. et al. Nickel, cobalt, and Ni-Co alloy nanopowders prepared by plasma-assisted milling: Catalytic activity in the carbon dioxide reforming of methane. Kinet Catal 55, 224–228 (2014). https://doi.org/10.1134/S0023158414020025
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158414020025