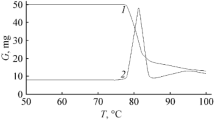
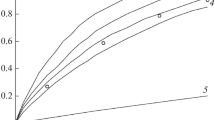
Abstract
The kinetics of 5-aminotetrazole thermal decomposition in the condensed phase at high heating rates (∼100 K/s) was studied by the dynamic mass spectrometric thermal analysis using a molecular beam sampling system, and the product composition was determined. Two routes of 5-aminotetrazole decomposition were distinguished, one yielding HN3 and NH2CN (route 1), and the other N2 and CH3N3 (route 2). The activation energy and rate constant of 5-AT decomposition were determined for each route.
Similar content being viewed by others
References
Kozyro, A.A., Simirskii, V.V., Krasulin, A.P., et al., Zh. Fiz. Khim., 1990, vol. 64, no. 3, p. 656.
Neidert, J.B., Black, R.E., Lynch, R.D., et al., Propulsion Conf., Cleveland, Ohio, 1998, vol. 2, p. 77.
Fallis, S., Reed, R., Lu, Y.-C., et al., Proc. Halon Options Technical Working Conf., Gaithersburg, Md.: NIST, 2000, p. 361.
Wierenga, P.H. and Holland, G.F., Proc. Halon Options Technical Working Conf., Gaithersburg Md.: NIST, 1999, p. 453.
Lesnikovich, A.I., Sviridov, V.V., Gaponik, P.N., et al., Dokl. Akad. Nauk BSSR, 1985, vol. 29, no. 3, p. 824.
Fogel’zang, A.E., Egorshev, V.Yu., Sinditskii, V.P., et al., Mater’yaly IX Vses. simp. po goreniyu i vzryvu (Proc. IX All-Union Symp. on Combustion and Explosion), Chernogolovka, Moscow oblast, 1989, p. 3.
Brill, T.B. and Ramanathan, H., Combust. Flame, 2000, vol. 122, p. 165.
Oyumi, G.Y. and Brill, T.B., Combust. Flame, 1991, vol. 83, p. 345.
Levchik, S.V., Ivashkevich, O.A., Balabanovich, A.I., et al., Thermochim. Acta, 1992, vol. 207, p. 115.
Lesnikovich, A.I., Ivashkevich, O.A., Levchik, S.V., et al., Thermochim. Acta, 2002, vol. 388, p. 233.
Brill, T.B., Prog. Energy Combust. Sci., 1992, vol. 18, p. 91.
Cronin, J.T. and Brill, T.B., Appl. Spectrosc., 1987, vol. 41, no. 7, p. 1147.
Jian-Guo Zhang., Li-Na Feng, Shao-Wen Zhang., and Tong-Lai Zhang., J. Mol. Model., 2008, no. 14, p. 403.
Chen, Z. and Xiao, H., Int. J. Quantum Chem., 2000, vol. 79, p. 350.
Shurukhin, Yu.V., Klyuev, N.A., and Grandberg, I.I., Khim. Geterotsikl. Soedin., 1982, no. 6, p. 723.
Reimlinger, H., Chem. Ind., 1972, p. 294.
Brady, L.E., J. Heterocycl. Chem., 1970, vol. 21, p. 1223.
Korobeinichev, O.P., Kuibida, L.V., Paletsky, A.A., and Shmakov, A.G., J. Propul. Power, 1998, vol. 14, no. 6, p. 991.
Korobeinichev, O.P., Fiz. Goreniya Vzryva, 1987, vol. 23, no. 5, p. 64.
US Patent 5594146, 1997.
Tereshenko, A.G., Korobeinichev, O.P., Paletsky, A.A., et al., Proc. 8th Int. Workshop on Combustion and Propulsion, Arzago d’Adda, Italy, 2002, p. 15.
NIST Chemical Webbook, http://webbook.nist.gov/chemistry/.
Rozenberg, A.S., Arsen’ev, Yu.N., and Voronkov, V.G., Fiz. Goreniya Vzryva, 1970, vol. 6, no. 3, p. 302.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Paletsky, N.V. Budachev, O.P. Korobeinichev, 2009, published in Kinetika i Kataliz, 2009, Vol. 50, No. 5, pp. 653–662.
Rights and permissions
About this article
Cite this article
Paletsky, A.A., Budachev, N.V. & Korobeinichev, O.P. Mechanism and kinetics of the thermal decomposition of 5-aminotetrazole. Kinet Catal 50, 627–635 (2009). https://doi.org/10.1134/S0023158409050036
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158409050036