Abstract
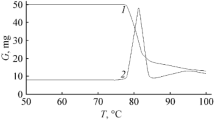
The thermal decomposition kinetics of 1-bromo-4-nitroxymethylcubane in the liquid phase is typical of C-ONO2 bond heterolysis, which occurs if the nitro ester has a strong donor substituent. A comparison between 1-bromo-4-nitroxymethylcubane and tert-butyl nitrate shows that bromocubyl is close to the tert-butyl group in induction properties and cubyl itself is a stronger donor than this group.
Similar content being viewed by others
References
Lur’e, B.A., Svetlov, B.S., and Shelaputina, V.P., Khimicheskaya fizika protsessov goreniya i vzryva. Kinetika khimicheskikh reaktsii (Chemical Physics of Combustion and Explosion. Chemical Kinetics), Chernogolovka, Moscow oblast: Otdelenie Inst. Khim. Fiz., 1977, p. 59.
Lurie, B., Tenth Symp. on Chemical Problems Connected with the Stability of Explosives, Margretetorp, 1955, p. 103.
Dubikhin, V.V., Lagodzinskaya, G.V., Korolev, A.M., Matveev, V.G., and Nazin, G.M., Khim. Fiz., 2003, vol. 22, p. 28.
Lur’e, B.A. and Svetlov, B.S., Kinet. Katal., 1994, vol. 35, no. 2, p. 165.
Kavell, D.N. and Sutthoff, A.F., J. Chem. Soc. B, 1969, no. 4, p. 366.
Prokudin, V.G., Lagodzinskaya, G.V., Dubikhin, V.V., Nazin, G.M., Romanova, L.B., and Eremenko, L.T., Kinet. Katal., 2005, vol. 45, no. 6, p. 847 [Kinet. Catal. (Engl. Transl.), vol. 45, no. 6, p. 800].
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.V. Dubikhin, V.G. Prokudin, L.D. Nazina, L.B. Romanova, L.T. Eremenko, G.M. Nazin, 2007, published in Kinetika i Kataliz, 2007, Vol. 48, No. 3, pp. 365–367.
Rights and permissions
About this article
Cite this article
Dubikhin, V.V., Prokudin, V.G., Nazina, L.D. et al. Kinetics and mechanism of the thermal decomposition of 1-bromo-4-nitroxymethylcubane. Kinet Catal 48, 345–347 (2007). https://doi.org/10.1134/S0023158407030020
Received:
Issue Date:
DOI: https://doi.org/10.1134/S0023158407030020