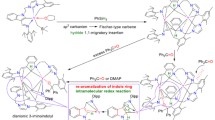
Abstract
New tin(IV) complexes based on various pyrocatechols containing tert-butyl and tert-octyl substituents in the aromatic ring are synthesized and structurally characterized. Reduction of spatially hindered o-quinones with tin amalgam in tetrahydrofuran allows the preparation of compounds with the trans-position of catecholate ligands in the metal coordination sphere. In the further reaction of synthesized tin(IV) biscatecholates with N-donor ligands (pyridine (Py), α,α′-dipyridyl (dipy), 1,10-phenanthroline (phen), pyrazino-[2,3-f][1-10]-phenanthroline (DPQ), dipyrido-[3,2-a:2′3′-c]-phenazine (DPPZ)) the respective six-coordinated complexes are obtained. The introduction of bidentate ligands naturally results in the cis-position of dioxolene ligands in the tin coordination sphere. The possibility of intramolecular ligand–ligand charge transfer (LL′CT) between catecholate and diimine moieties in the synthesized octahedral metal derivatives is shown by electronic absorption spectroscopy. The effect of substituents in catecholate ligands and acceptor properties of diimines on the shift of a long-wave absorption band is determined. The electronic transition corresponding to LL′CT is revealed to undergo a bathochromic shift in the electronic absorption spectrum with a decrease in the polarity of the solvent used.














Similar content being viewed by others
REFERENCES
C. Pierpont. Unique properties of transition metal quinone complexes of the MQ3 series. Coord. Chem. Rev., 2001, 219-221, 415-433. https://doi.org/10.1016/s0010-8545(01)00342-3
M. P. Bubnov, A. V. Piskunov, A. A. Zolotukhin, I. N. Meshcheryakova, N. A. Skorodumova, A. S. Bogomyakov, E. V. Baranov, G. K. Fukin, and V. K. Cherkasov. Homoligand tris-o-dioxolene complexes. Peculiarities of the molecular structures and magnetic properties. Russ. J. Coord. Chem., 2020, 46(4), 224-240. https://doi.org/10.1134/s107032842003001x
I. V. Ershova and A. V. Piskunov. Complexes of group III metals based on o-iminoquinone ligands. Russ. J. Coord. Chem., 2020, 46(3), 154-177. https://doi.org/10.1134/s1070328420030021
A. Rajput, A. K. Sharma, S. K. Barman, A. Saha, and R. Mukherjee. Valence tautomerism and delocalization in transition metal complexes of o-amidophenolates and other redox-active ligands. Some recent results. Coord. Chem. Rev., 2020, 414, 213240. https://doi.org/10.1016/j.ccr.2020.213240
K. I. Pashanova, A. I. Poddel′sky, and A. V. Piskunov. Complexes of ″late″ transition metals of the 3d row based on functionalized o-iminobenzoquinone type ligands: Interrelation of molecular and electronic structure, magnetic behaviour. Coord. Chem. Rev., 2022, 459, 214399. https://doi.org/10.1016/j.ccr.2021.214399
M. G. Chegerev and A. V. Piskunov. Chemistry of complexes of group 14 elements based on redox-active ligands of the o-iminoquinone type. Russ. J. Coord. Chem., 2018, 44(4), 258-271. https://doi.org/10.1134/s1070328418040036
W. Kaim, A. Das, J. Fiedler, S. Záliš, and B. Sarkar. NO and NO2 as non-innocent ligands: A comparison. Coord. Chem. Rev., 2020, 404, 213114. https://doi.org/10.1016/j.ccr.2019.213114
I. V. Ershova, A. V. Piskunov, and V. K. Cherkasov. Complexes of diamagnetic cations with radical anion ligands. Russ. Chem. Rev., 2020, 89(11), 1157-1183. https://doi.org/10.1070/rcr4957
R. M. Buchanan and C. G. Pierpont. Tautomeric catecholate-semiquinone interconversion via metal-ligand electron transfer. Structural, spectral, and magnetic properties of (3,5-di-tert-butylcatecholato)(3,5-di-tert-butylsemiquinone)(bipyridyl)cobalt(III), a complex containing mixed-valence. J. Am. Chem. Soc., 1980, 102(15), 4951-4957. https://doi.org/10.1021/ja00535a021
C. G. Pierpont. Studies on charge distribution and valence tautomerism in transition metal complexes of catecholate and semiquinonate ligands. Coord. Chem. Rev., 2001, 216/217, 99-125. https://doi.org/10.1016/s0010-8545(01)00309-5
T. Tezgerevska, K. G. Alley, and C. Boskovic. Valence tautomerism in metal complexes: Stimulated and reversible intramolecular electron transfer between metal centers and organic ligands. Coord. Chem. Rev., 2014, 268, 23-40. https://doi.org/10.1016/j.ccr.2014.01.014
M. P. Bubnov, N. A. Skorodumova, G. K. Fukin, R. V. Rumyantsev, A. A. Zolotukhin, A. S. Bogomyakov, and V. K. Cherkasov. .Solid solutions of redox-isomeric bis-o-semiquinonato cobalt complex with zinc, nickel and manganese compounds having the same composition. Polyhedron, 2021, 209, 115485. https://doi.org/10.1016/j.poly.2021.115485
S. O. Shapovalova, A. A. Guda, M. P. Bubnov, G. Smolentsev, Y. V. Rusalev, V. V. Shapovalov, A. A. Zolotukhin, V. K. Cherkasov, A. G. Starikov, V. G. Vlasenko, and A. V. Soldatov. Temperature and time-resolved XANES studies of novel valence tautomeric cobalt complex. Chem. Lett., 2021, 50(11), 1933-1937. https://doi.org/10.1246/cl.210426
V. K. Cherkasov, G. A. Abakumov, E. V. Grunova, A. I. Poddel′sky, G. K. Fukin, E. V. Baranov, Y. V. Kurskii, and L. G. Abakumova. Triphenylantimony(V) catecholates and o-amidophenolates: Reversible binding of molecular oxygen. Chem. - Eur. J., 2006, 12(14), 3916-3927. https://doi.org/10.1002/chem.200501534
K. I. Pashanova, V. O. Bitkina, I. A. Yakushev, M. V. Arsenyev, and A. V. Piskunov. Square-planar heteroleptic complexes of α-diimine-NIII-catecholate type: Intramolecular ligand-to-ligand charge transfer. Molecules, 2021, 26(15), 4622. https://doi.org/10.3390/molecules26154622
A. V. Maleeva, I. V. Ershova, O. Y. Trofimova, K. V. Arsenyeva, I. A. Yakushev, and A. V. Piskunov. Near-IR absorbing donor–acceptor charge-transfer gallium complex, an example from non-transition metal chemistry. Mendeleev Commun., 2022, 32(1), 83-86. https://doi.org/10.1016/j.mencom.2022.01.027
I. V. Ershova, A. V. Maleeva, R. R. Aysin, A. V. Cherkasov, and A. V. Piskunov. Effect of crystal packing on charge transfer in the heteroleptic gallium(III) complex. Russ. Chem. Bull., 2023, 72(1), 193-201. https://doi.org/10.1007/s11172-023-3724-2
A. V. Maleeva, O. Y. Trofimova, I. V. Ershova, K. V. Arsenyeva, K. I. Pashanova, I. A. Yakushev, A. V. Cherkasov, R. R. Aysin, and A. V. Piskunov. Molecular and electronic structures of paramagnetic gallium complexes with differently charged o-quinone ligands. Russ. Chem. Bull., 2022, 71(7), 1441-1452. https://doi.org/10.1007/s11172-022-3550-y
L. A. Cameron, J. W. Ziller, and A. F. Heyduk. Near-IR absorbing donor–acceptor ligand-to-ligand charge-transfer complexes of nickel(II). Chem. Sci., 2016, 7(3), 1807-1814. https://doi.org/10.1039/c5sc02703a
W. W. Kramer, L. A. Cameron, R. A. Zarkesh, J. W. Ziller, and A. F. Heyduk. Donor–acceptor ligand-to-ligand charge-transfer coordination complexes of nickel(II). Inorg. Chem., 2014, 53(16), 8825-8837. https://doi.org/10.1021/ic5017214
S. Archer and J. A. Weinstein. Charge-separated excited states in platinum(II) chromophores: Photophysics, formation, stabilization and utilization in solar energy conversion. Coord. Chem. Rev., 2012, 256(21/22), 2530-2561. https://doi.org/10.1016/j.ccr.2012.07.010
M. O. BaniKhaled, J. D. Becker, M. Koppang, and H. Sun. Perfluoroalkylation of square-planar transition metal complexes: A strategy to assemble them into solid state materials with a π–π stacked lamellar structure. Cryst. Growth Des., 2016, 16(4), 1869-1878. https://doi.org/10.1021/acs.cgd.5b01291
T. Thorwart, D. Hartmann, and L. Greb. Dihydrogen activation with a neutral, intermolecular silicon(IV)-amine frustrated Lewis pair. Chem. - Eur. J., 2022, 28(60). https://doi.org/10.1002/chem.202202273
T. Thorwart, D. Roth, and L. Greb. Bis(pertrifluoromethylcatecholato)silane: Extreme Lewis acidity broadens the catalytic portfolio of silicon. Chem. - Eur. J., 2021, 27(40), 10422-10427. https://doi.org/10.1002/chem.202101138
D. Hartmann, S. Braner, and L. Greb. Bis(perchlorocatecholato)silane and heteroleptic bidonors: Hidden frustrated Lewis pairs resulting from ring strain. Chem. Commun., 2021, 57(69), 8572-8575. https://doi.org/10.1039/d1cc03452a
N. Ansmann, T. Thorwart, and L. Greb. Silicon catalyzed C–O bond ring closing metathesis of polyethers. Angew. Chem., 2022, 134(44). https://doi.org/10.1002/ange.202210132
K. V. Arsenyeva, K. I. Pashanova, O. Y. Trofimova, I. V. Ershova, M. G. Chegerev, A. A. Starikova, A. V. Cherkasov, M. A. Syroeshkin, A. Y. Kozmenkova, and A. V. Piskunov. O,N-heterocyclic germylenes as efficient catalysts for hydroboration and cyanosilylation of benzaldehyde. New J. Chem., 2021, 45(26), 11758-11767. https://doi.org/10.1039/d1nj01644j
K. V. Arsenyeva, A. V. Klimashevskaya, K. I. Pashanova, O. Y. Trofimova, M. G. Chegerev, A. A. Starikova, A. V. Cherkasov, G. K. Fukin, I. A. Yakushev, and A. V. Piskunov. Stable heterocyclic stannylene: The metal, ligand-centered reactivity, and effective catalytic hydroboration of aldehydes. Appl. Organomet. Chem., 2022, 36(4). https://doi.org/10.1002/aoc.6593
L. B. Vaganova, A. V. Maleeva, A. V. Piskunov, and D. F. Grishin. Bis-catecholate complexes of the IV group elements in the radical polymerization of styrene: ESR studies. Russ. Chem. Bull., 2011, 60(8), 1620-1627. https://doi.org/10.1007/s11172-011-0242-4
E. V. Kolyakina, L. B. Vaganova, A. V. Lado, A. V. Piskunov, V. K. Cherkasov, and D. F. Grishina. Bis(3,6-di-tert-butylcatecholato)tin(IV) ditetrahydrofuranate in radical polymerization of methyl methacrylate. Russ. Chem. Bull., 2007, 56(7), 1363-1368. https://doi.org/10.1007/s11172-007-0208-8
A. V. Piskunov, O. Y. Trofimova, M. S. Piskunova, I. V. Smolyaninov, N. T. Berberova, and G. K. Fukin. Template assembling of the pentadentate redox-active ligand in the coordination sphere of tin(IV). Russ. J. Coord. Chem., 2018, 44(2), 138-146. https://doi.org/10.1134/s1070328418020082
A. V. Piskunov, O. Y. Trofimova, A. V. Maleeva, and A. V. Cherkasov. Template synthesis of tin(IV) complexes with tridentate iminopyridine ligands. Russ. J. Coord. Chem., 2019, 45(3), 188-199. https://doi.org/10.1134/s1070328419020040
E. N. Nikolaevskaya, E. A. Saverina, A. A. Starikova, A. Farhati, M. A. Kiskin, M. A. Syroeshkin, M. P. Egorov, and V. V. Jouikov. Halogen-free GeO2 conversion: Electrochemical reduction vs. complexation in (DTBC)2Ge[Py(CN)n] (n = 0 … 2) complexes. Dalton Trans., 2018, 47(47), 17127-17133. https://doi.org/10.1039/c8dt03397h
P. G. Shangin, I. V. Krylova, A. V. Lalov, A. Y. Kozmenkova, E. A. Saverina, P. A. Buikin, A. A. Korlyukov, A. A. Starikova, E. N. Nikolaevskaya, M. P. Egorov, and M. A. Syroeshkin. Supramolecular D⋯A-layered structures based on germanium complexes with 2,3-dihydroxynaphthalene and N,N′-bidentate ligands. RSC Adv., 2021, 11(35), 21527-21536. https://doi.org/10.1039/d1ra02691g
I. V. Krylova, L. D. Labutskaya, M. O. Markova, V. A. Balycheva, P. G. Shangin, A. Y. Akyeva, V. V. Golovina, M. E. Minyaev, A. V. Lalov, V. M. Pechennikov, V. T. Novikov, M. P. Egorov, and M. A. Syroeshkin. Derivatives of penta-, hexa-, and hepta-coordinated tin with Schiff bases and 1,10-phenanthroline: Structure, redox and optoelectronic properties. New J. Chem., 2023, 47(25), 11890-11902. https://doi.org/10.1039/d3nj01993d
A. F. Akbulatov, A. Y. Akyeva, P. G. Shangin, N. A. Emelianov, I. V. Krylova, M. O. Markova, L. D. Labutskaya, A. V. Mumyatov, E. I. Tuzharov, D. A. Bunin, L. A. Frolova, M. P. Egorov, M. A. Syroeshkin, and P. A. Troshin. Sn and Ge complexes with redox-active ligands as efficient interfacial membrane-like buffer layers for p-i-n perovskite solar cells. Membranes, 2023, 13(4), 439. https://doi.org/10.3390/membranes13040439
R. M. Clarke, K. Hazin, J. R. Thompson, D. Savard, K. E. Prosser, and T. Storr. Electronic structure description of a doubly oxidized bimetallic cobalt complex with proradical ligands. Inorg. Chem., 2016, 55(2), 762-774. https://doi.org/10.1021/acs.inorgchem.5b02231
L. Chiang, K. Herasymchuk, F. Thomas, and T. Storr. Influence of electron-withdrawing substituents on the electronic structure of oxidized Ni and Cu salen complexes. Inorg. Chem., 2015, 54(12), 5970-5980. https://doi.org/10.1021/acs.inorgchem.5b00783
K. I. Pashanova, I. V. Ershova, O. Y. Trofimova, R. V. Rumyantsev, G. K. Fukin, A. S. Bogomyakov, M. V. Arsenyev, and A. V. Piskunov. Charge transfer chromophores derived from 3d-row transition metal complexes. Molecules, 2022, 27(23), 8175. https://doi.org/10.3390/molecules27238175
A. V. Maleeva, O. Y. Trofimova, I. A. Yakushev, R. R. Aysin, and A. V. Piskunov. Heteroleptic zinc catecholate complexes with N-donor ligands. Russ. J. Coord. Chem., 2023, 49(7), 420-428. https://doi.org/10.1134/s1070328423600134
D. D. Perrin, W. L. F. Armarego, and D. R. Perrin. Purification of laboratory chemicals. Oxford, UK: Pergamon, 1980.
M. A. Ivanov, M. V. Puzyk, and K. P. Balashev. Spectroscopic and electrochemical properties of dichlorodiimine complexes of Au(III) and Pt(II) with 1,4-diazine derivatives of o-phenanthroline. Russ. J. Gen. Chem., 2006, 76(6), 843-848. https://doi.org/10.1134/s1070363206060016
M. R. Waterland, K. C. Gordon, J. J. McGarvey, and P. M. Jayaweera. Spectroscopic and electrochemical studies of a series of copper(I) and rhenium(I) complexes with substituted dipyrido[3,2-a:2′,3′-c]phenazine ligands. J. Chem. Soc., Dalton Trans., 1998, (4), 609-616. https://doi.org/10.1039/a706110b
W. Flaig, T. Ploetz, and H. Biergans. Zur Kenntnis der Huminsäuren, XIV. Mitteilung Bildung und Reaktionen einiger Hydroxy-chinone. Justus Liebigs Ann. Chem., 1955, 597(3), 196-213. https://doi.org/10.1002/jlac.19555970304
T. N. Kocherova, N. O. Druzhkov, M. V. Arsenyev, E. V. Baranov, V. A. Kuropatov, and V. K. Cherkasov. New o-benzoquinones substituted with tert-octyl groups. Russ. Chem. Bull., 2023, 72(5), 1192-1202. https://doi.org/10.1007/s11172-023-3889-8
G. Brauer. Handbuch der Praparativen Anorganishen Chemie, 3rd ed. Stuttgard, Germany: Ferdinand Enke, 1981.
R. D. Svetogorov, P. V. Dorovatovskii, and V. A. Lazarenko. Belok/XSA diffraction beamline for studying crystalline samples at kurchatov synchrotron radiation source. Cryst. Res. Technol., 2020, 55(5). https://doi.org/10.1002/crat.201900184
W. Kabsch. XDS. Acta Crystallogr., Sect. D: Biol. Crystallogr., 2010, 66(2), 125-132. https://doi.org/10.1107/s0907444909047337
CrysAlisPro software system, ver. 1.171.35.19. Wroclaw, Poland: Rigaku Corporation, 2022.
APEX3, SAINT, and SADABS. Madison, Wisconsin, USA Bruker, AXS and Inc., 2016.
G. M. Sheldrick. SHELXT - Integrated space-group and crystal-structure determination. Acta Crystallogr., Sect. A: Found. Adv., 2015, 71(1), 3-8. https://doi.org/10.1107/s2053273314026370
G. M. Sheldrick. Crystal structure refinement with SHELXL. Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71(1), 3-8. https://doi.org/10.1107/s2053229614024218
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. Puschmann. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr., 2009, 42(2), 339-341. https://doi.org/10.1107/s0021889808042726
A. V. Piskunov, A. V. Lado, G. K. Fukin, E. V. Baranov, L. G. Abakumova, V. K. Cherkasov, and G. A. Abakumov. The reaction of 3,6-di-tert-butyl-o-benzoquinone with tin amalgam: Synthesis and structure of tin catecholato complexes. Heteroat. Chem., 2006, 17(6), 481-490. https://doi.org/10.1002/hc.20271
A. V. Yatsenko, L. A. Aslanov, M. Yu. Burtsev, and E. A. Kravchenko. Crystalline and molecular-structure and Cl-35 n.q.r. spectra of bis-(diethylesterate)-tetrachlorotin SnCl4(Et2O)2. Russ. J. Inorg. Chem., 1991, 36, 8, 2031.
T. A. Annan, B. R. McGarvey, A. Ozarowski, D. G. Tuck, and R.K. Chadha. One-electron transfer processes in the reaction of tin(II) halides with substituted o-quinones; crystal structure of bis(3,5-di-t-butylcatecholato)tin–1,10-phenanthroline–dimethylformamide (1/1/2). J. Chem. Soc., Dalton Trans., 1989, (3), 439-446. https://doi.org/10.1039/dt9890000439
S. N. Brown. Metrical oxidation states of 2-amidophenoxide and catecholate ligands: structural signatures of metal–ligand π bonding in potentially noninnocent ligands. Inorg. Chem., 2012, 51(3), 1251-1260. https://doi.org/10.1021/ic202764j
A. Asadi, C. Eaborn, M. S. Hill, P. B. Hitchcock, M. M. Meehan, and J. D. Smith. Reactions of a highly crowded cyclic stannylene with iodoalkanes, enones, and dienes. Inhibition of nucleophilic substitution at tin(IV) centers. Organometallics, 2002, 21(12), 2430-2437. https://doi.org/10.1021/om020106y
S. V. Baryshnikova, A. I. Poddel′sky, E. V. Bellan, I. V. Smolyaninov, A. V. Cherkasov, G. K. Fukin, N. T. Berberova, V. K. Cherkasov, and G. A. Abakumov. Ferrocene-containing tin(IV) complexes based on o-benzoquinone and o-iminobenzoquinone ligands. Synthesis, molecular structure, and electrochemical properties. Inorg. Chem., 2020, 59(10), 6774-6784. https://doi.org/10.1021/acs.inorgchem.9b03757
A. V. Piskunov, A. V. Lado, E. V. Ilyakina, G. K. Fukin, E. V. Baranov, V. K. Cherkasov, and G. A. Abakumov. New organobimetallic compounds containing catecholate and o-semiquinolate ligands. J. Organomet. Chem., 2008, 693(1), 128-134. https://doi.org/10.1016/j.jorganchem.2007.10.029
C. Reichardt and T. Welton. Solvents and Solvent Effects in Organic Chemistry. Wiley, 2010. https://doi.org/10.1002/9783527632220
Funding
The work was supported by the Grant Council of the President of the Russian Foundation for supporting the leading scientific schools (grant No. Nsh-403.2022.1.3). Works on structural studies of compounds 5b and 7d were supported by the Ministry of Education of the Russian Foundation within the State Assignment for the Institute of General and Inorganic Chemistry, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors of this work declare that they have no conflicts of interests.
Additional information
Russian Text © The Author(s), 2023, published in Zhurnal Strukturnoi Khimii, 2023, Vol. 64, No. 12, 118910.https://doi.org/10.26902/JSC_id118910
Publisher’s Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Klimashevskaya, A.V., Arsenyeva, K.V., Cherkasov, A.V. et al. Tin(IV) Complexes Based on Diimines and 3,5-Di-Tert-Alkyl Substituted Pyrocatechols. J Struct Chem 64, 2271–2294 (2023). https://doi.org/10.1134/S0022476623120016
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476623120016